Home PageAbout MindatThe Mindat ManualHistory of MindatCopyright StatusWho We AreContact UsAdvertise on Mindat
Donate to MindatCorporate SponsorshipSponsor a PageSponsored PagesMindat AdvertisersAdvertise on Mindat
Learning CenterWhat is a mineral?The most common minerals on earthInformation for EducatorsMindat ArticlesThe ElementsThe Rock H. Currier Digital LibraryGeologic Time
Minerals by PropertiesMinerals by ChemistryAdvanced Locality SearchRandom MineralRandom LocalitySearch by minIDLocalities Near MeSearch ArticlesSearch GlossaryMore Search Options
The Mindat ManualAdd a New PhotoRate PhotosLocality Edit ReportCoordinate Completion ReportAdd Glossary Item
Mining CompaniesStatisticsUsersMineral MuseumsClubs & OrganizationsMineral Shows & EventsThe Mindat DirectoryDevice SettingsThe Mineral Quiz
Photo SearchPhoto GalleriesSearch by ColorNew Photos TodayNew Photos YesterdayMembers' Photo GalleriesPast Photo of the Day GalleryPhotography
╳Discussions
💬 Home🔎 Search📅 LatestGroups
EducationOpen discussion area.Fakes & FraudsOpen discussion area.Field CollectingOpen discussion area.FossilsOpen discussion area.Gems and GemologyOpen discussion area.GeneralOpen discussion area.How to ContributeOpen discussion area.Identity HelpOpen discussion area.Improving Mindat.orgOpen discussion area.LocalitiesOpen discussion area.Lost and Stolen SpecimensOpen discussion area.MarketplaceOpen discussion area.MeteoritesOpen discussion area.Mindat ProductsOpen discussion area.Mineral ExchangesOpen discussion area.Mineral PhotographyOpen discussion area.Mineral ShowsOpen discussion area.Mineralogical ClassificationOpen discussion area.Mineralogy CourseOpen discussion area.MineralsOpen discussion area.Minerals and MuseumsOpen discussion area.PhotosOpen discussion area.Techniques for CollectorsOpen discussion area.The Rock H. Currier Digital LibraryOpen discussion area.UV MineralsOpen discussion area.Recent Images in Discussions
Identity HelpWhite Erythrite?
28th May 2012 02:01 UTCReiner Mielke Expert
28th May 2012 08:39 UTCPeter Haas
Since Ni2+ has the lower specific absorptivity, there can be white and even pink annabergite (to compensate the colour produced by a given amount of Ni2+, it takes a lesser amount of Co2+). For the same reason however, colourless erxthrite does not exist (it would take more Ni2+ than Co2+, which places the specimen in the compositional range of annabergite).
In aqueous HCl, the situation is a similar one in that the different species (chlorocobaltate and chloronickelate, respectively) have different specific absorptivities. Although their colours will not cancel, the colour of one or the other will be dominant, depending on concentration. In addition, the colour of the cobalt-bearing solution will gradually change from blue to pale pink with decreasing temperature and concentration (this is an equilibrium effect between two different species; you don't have either one or the other, but always both of them in varying proportions). While the pink aquo-complex is a weak chromophor (i.e. its specific absorptivity is relatively low), the blue chloro-complex is a strong chromophor. Thus, temperature and concentration are another two important parameters to consider when conclusions have to be drawn from the colour of the solution. The only logical conclusion however is that this test is not conclusive unless the specimen has a near end-member composition of either erythrite or annabergite.

28th May 2012 09:57 UTCUwe Ludwig
Uwe Ludwig
28th May 2012 16:31 UTCReiner Mielke Expert

28th May 2012 16:49 UTCLefteris Rantos Expert
EDS should clearly differentiate between a Vivianite-group mineral containing some of the above elements, Pharmacolite (with Ca) and Picropharmacolite (with Ca and Mg).
Lefteris.
22nd Jun 2012 01:19 UTCReiner Mielke Expert
22nd Jun 2012 04:34 UTCPeter Haas
23rd Jun 2012 03:24 UTCPeter Nancarrow 🌟 Expert
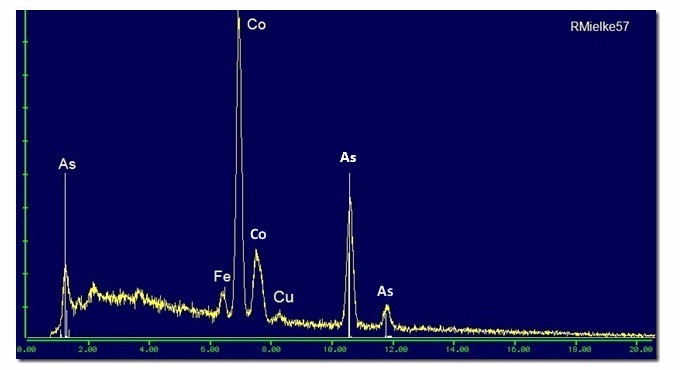
The energy of Cu Kα is 8.04 KeV, whereas that peak is actually nearer 8.25 KeV, which is about right for NiKβ.
Therefore the peak at about 7.5 KeV labelled "Co", is an overlapping composite of NiKα (7.47 KeV) and Co Kβ (7.65 KeV) and there is no significant Cu in the specimen.
Pete N.
24th Jun 2012 14:23 UTCReiner Mielke Expert
Thank you! I didn't do the EDS but will contact the person who did. A high nickel content could account for the color.

24th Jun 2012 16:30 UTCKerry Day
25th Jun 2012 01:53 UTCReiner Mielke Expert

25th Jun 2012 02:24 UTCAlfredo Petrov Manager
26th Jun 2012 01:03 UTCReiner Mielke Expert
Have never heard of that happening. You find eyrthrite on dumps that has been in the sun for years that is bright pink, besides this was found buried deep in moist rubble and it came out white.
26th Jun 2012 16:52 UTCUwe Kolitsch Manager
It appears, however, there is not much Mg.




Mindat.org is an outreach project of the Hudson Institute of Mineralogy, a 501(c)(3) not-for-profit organization.
Copyright © mindat.org and the Hudson Institute of Mineralogy 1993-2024, except where stated. Most political location boundaries are © OpenStreetMap contributors. Mindat.org relies on the contributions of thousands of members and supporters. Founded in 2000 by Jolyon Ralph.
Privacy Policy - Terms & Conditions - Contact Us / DMCA issues - Report a bug/vulnerability Current server date and time: April 23, 2024 07:10:30
Copyright © mindat.org and the Hudson Institute of Mineralogy 1993-2024, except where stated. Most political location boundaries are © OpenStreetMap contributors. Mindat.org relies on the contributions of thousands of members and supporters. Founded in 2000 by Jolyon Ralph.
Privacy Policy - Terms & Conditions - Contact Us / DMCA issues - Report a bug/vulnerability Current server date and time: April 23, 2024 07:10:30