Home PageAbout MindatThe Mindat ManualHistory of MindatCopyright StatusWho We AreContact UsAdvertise on Mindat
Donate to MindatCorporate SponsorshipSponsor a PageSponsored PagesMindat AdvertisersAdvertise on Mindat
Learning CenterWhat is a mineral?The most common minerals on earthInformation for EducatorsMindat ArticlesThe ElementsThe Rock H. Currier Digital LibraryGeologic Time
Minerals by PropertiesMinerals by ChemistryAdvanced Locality SearchRandom MineralRandom LocalitySearch by minIDLocalities Near MeSearch ArticlesSearch GlossaryMore Search Options
The Mindat ManualAdd a New PhotoRate PhotosLocality Edit ReportCoordinate Completion ReportAdd Glossary Item
Mining CompaniesStatisticsUsersMineral MuseumsClubs & OrganizationsMineral Shows & EventsThe Mindat DirectoryDevice SettingsThe Mineral Quiz
Photo SearchPhoto GalleriesSearch by ColorNew Photos TodayNew Photos YesterdayMembers' Photo GalleriesPast Photo of the Day GalleryPhotography
╳Discussions
💬 Home🔎 Search📅 LatestGroups
EducationOpen discussion area.Fakes & FraudsOpen discussion area.Field CollectingOpen discussion area.FossilsOpen discussion area.Gems and GemologyOpen discussion area.GeneralOpen discussion area.How to ContributeOpen discussion area.Identity HelpOpen discussion area.Improving Mindat.orgOpen discussion area.LocalitiesOpen discussion area.Lost and Stolen SpecimensOpen discussion area.MarketplaceOpen discussion area.MeteoritesOpen discussion area.Mindat ProductsOpen discussion area.Mineral ExchangesOpen discussion area.Mineral PhotographyOpen discussion area.Mineral ShowsOpen discussion area.Mineralogical ClassificationOpen discussion area.Mineralogy CourseOpen discussion area.MineralsOpen discussion area.Minerals and MuseumsOpen discussion area.PhotosOpen discussion area.Techniques for CollectorsOpen discussion area.The Rock H. Currier Digital LibraryOpen discussion area.UV MineralsOpen discussion area.Recent Images in Discussions
Identity HelpBasement Minerals
17th Aug 2011 20:02 UTCReiner Mielke Expert
17th Aug 2011 20:41 UTCJolyon Ralph Founder
sadly not by current official definitions. But how you choose to classify it is entirely up to you! Nice find.
23rd Aug 2011 18:42 UTCRob Woodside 🌟 Manager
23rd Aug 2011 23:22 UTCReiner Mielke Expert

29th Aug 2011 03:26 UTCHoward Heitner
29th Aug 2011 14:23 UTCReiner Mielke Expert
30th Aug 2011 02:10 UTCPaul Brandes 🌟 Manager
When I saw "Basement Minerals", first thing I thought of was the crystalline rocks/minerals of the Canadian Shield..... B)

30th Aug 2011 09:37 UTCHoward Heitner
30th Aug 2011 19:19 UTCRob Woodside 🌟 Manager
We seem to be starting a new geologic age with human intervention and when this is accepted maybe the IMA will accept anthropogenic artifacts as minerals.

30th Aug 2011 21:20 UTCUwe Ludwig
Uwe Ludwig
31st Aug 2011 09:26 UTCUwe Kolitsch Manager

31st Aug 2011 12:32 UTCTomas Husdal Expert
I will, on purpose, place boulders rich in REE-minerals on the seashore, and return in some years to look for new minerals with REE, Na and Cl...

31st Aug 2011 19:01 UTCUwe Ludwig
Uwe Ludwig

31st Aug 2011 19:34 UTCEvan Johnson

3rd Sep 2011 09:33 UTCUwe Ludwig
Uwe Ludwig
3rd Sep 2011 15:09 UTCReiner Mielke Expert
Great idea, I think I will start a mineral garden in my backyard.

3rd Sep 2011 16:30 UTCSpencer Ivan Mather
email address.........garnets@hotmail.co.uk
Thank you. Spencer.

3rd Sep 2011 16:53 UTCAlfredo Petrov Manager
But since you've opened it and exposed it to the air, it will get destroyed - Seal it up again, quick!
3rd Sep 2011 17:10 UTCReiner Mielke Expert
Take a picture so even if it gets destroyed you still have a record of it, would love to see it.
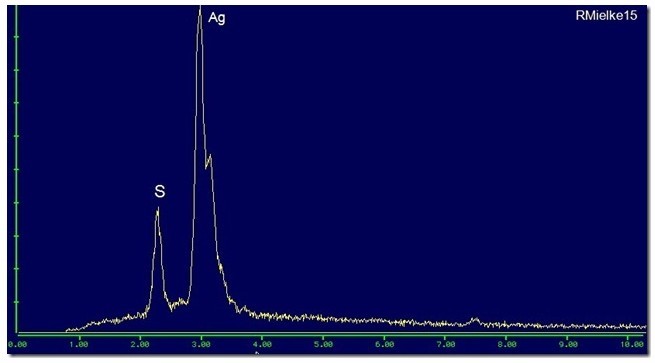
21st Sep 2011 01:49 UTCReiner Mielke Expert
21st Sep 2011 20:00 UTCReiner Mielke Expert

21st Sep 2011 21:49 UTCBart Cannon
Materials science people call the process "gettering".
Copper and silver draw sulfur out of the atmosphere, and especially so in a partial vacuum.
If I leave metallic copper or silver in the probe for a few days those metals get coated with sulfides.
Tarnish on silver teapots might have the acicular habit if air currents around the house weren't killing the delicate structures.
I would agree with Rob that acanthite fuzz is not a mineral, but please bring back the Laurium minerals to full status. Human beings are 100% natural creatures. Just like bacteria which are responsible for the formation of most of the secondary minerals we all love.
I draw the line at engineered materials such as alloys. But I think this whole area of discourse is kind of wobbly.
I have seen beautiful crystals of synthetic quartz, crocoite, anglesite etc. Them things are minerals.
Mandarino is dead. Can't we reverse his decision on Laurium? With a respectful footnote ?
21st Sep 2011 22:52 UTCReiner Mielke Expert

21st Sep 2011 23:52 UTCBart Cannon
I wonder if there could be a little native silver intergrown with the arsenides.
This could be a new category of minerals to collect and curate.
There are a host of sulfates that grow on pyrrhotite and marcasite in the drawers. Some beautiful and delicate.
My favorite off center collecting category is "mineral creations".
My first experience was chalcanthite. No glue needed. No artistic skill needed either, so I put them in the "Low" categorty.
I have a fantastic "created" leadhillite from Tiger, AZ. It is a PERFECT thumbnail. I will never sell it, and I will never sell my texasites.

22nd Sep 2011 11:39 UTCBart Cannon
I can't find any on simple native copper or native silver specimens. Just tarnish.
The only time I see them is on specimens with silver bearing minerals associated with other sulfides or sulfiur bearing selenides.
Could Thiobacillus (bacteria) be involved ? Bacteria need moisture. Is acanthite fuzz more prevalent in moist climates ?
One of my first commercial accounts was with Sunstrand Data and Allied Signal formerly of Redmond, Washington. They brought me their failed $8,000 miniature accelerometers. The same acceleromters used in the Sea Snake missiles that took down that Iranian passenger jet long ago.
The accelerometers were a wonder of technoloty, but kept failing because of embrittlement at the lead pad / copper coil wire joint on the thin quartz reed. The mechanism was in a vacuum, and extremely elegant. The lead pads all showed tiny arborscent copper sulfide crystals. The O-Rings used to seal the assembly contained 6% sulfur. I suggested they used a low sulfur polyurethane O-ring, but 15 years later my last job looked just like the first, and they were still using sulfur bearing O-rings. I could never figure out why they bothered to hire me for at least 100 analyses.
I had an engineer friend at Boeing whose job was to perfect the growth of tungsten "whiskers". That sort of thing is now part of the trendy buzz term known as nanotechnology. I wonder if acanthite whiskers might become a big play in that field.
22nd Sep 2011 13:04 UTCReiner Mielke Expert
The sample I had was in an egg carton with 11 other silver samples none of which had any fuzz. Don't know what made the one different from the others other than it was the only one with a bit of arsenide. Must require some very unique conditions to grow.
"I could never figure out why they bothered to hire me for at least 100 analyses." Guess they just wanted to know why they failed and not how to correct the problem. But then maybe it isn't a problem, I mean they could make cars that don't rust but they don't because there is more money to made with cars that do rust. Such is the world of capitalism.

22nd Sep 2011 15:42 UTCSpencer Ivan Mather
Spencer.
22nd Sep 2011 22:49 UTCVandall Thomas King Manager

24th Sep 2011 15:50 UTCDana Morong
Chemical News, 1877, volume 35:
Feb. 16, pages 68-71 On the Formation of Moss Gold and Silver
(Archibald Liversidge)
March 23, pp. 117-118 On the Formation of “Moss Copper” (W. M. Hutchings)
April 6, pp. 144-145 Moss Copper, &c. (T. A. Readwin, letter)
April 13, p. 154. Moss Copper, &c. (J. H. Collins)
May 4, pp.180-182 Formation of Moss Copper, &c. (W. M. Hutchings)
May 4, pp. 186-187 Moss-Copper, &c. (T. A. Readwin)
May 11, pp. 195-196 Formation of Moss-Copper, &c. (T. A. Readwin)
Mining and Metallurgy, 1907, Amer. Inst. of M., M., P. E., p. 214
“Growing Wire Silvers” letter by Don Edwards, of Tideswell, UK, in Letters, in Mineralogical Record, January-February 2001, v.32, #1, 72-73.
“On a Fibrous Metallic Copper” by James Smithson, 1820, from Thomson’s Annals of Philosophy, Vol. XVI, 1820, p. 46, and reprinted in The Scientific Writings of James Smithson, edited by William J. Rhees, 1879, in Smithsonian Miscellaneous Collections, #327, in volume 21, p. 68-71.
“On some Capillary Metallic Tin” by James Smithson, 1821, from Thomson’s Annals of Philosophy, Vol. XII, 1821, or New Series Vol. I, p.271, reprinted in The Scientific Writings of James Smithson, edited by William J. Rhees, 1879, in Smithsonian Miscellaneous Collections, #327, in volume 21, p.75.
There are other references which I have not accessed yet:
Principles of Extractive Metallurgy, Fathi Habashi, Volume 3: Pyrometallurgy, 1969, page 201
Metallurgy: The Art of Extracting Metals from their Ores (John Percy, 1861, 596 pages,
(moss copper, pp. 342-350... & beyond)
The Ore Deposits of the United States and Canada (James Furman Kemp, 1900, 481 pages)
Handbook of Metallurgy, Carl Schnabel, Henry Louis, 1898
I do not know whether Ramdohr’s large volume The Ore Minerals and Their Intergrowths would have any mention of this phenomenon, although I doubt it, as this book is fairly recent, and the phenomenon attracted attention mainly many decades ago; it does not seem to attract academic attention now, perhaps because it has too little appeal to grantors of graduate school projects, and also possibly because there is doubt about the growth’s status as minerals. One of the most recent mentions, and the most accessible, is the letter (mentioned) in Mineralogical Record in 2001 (volume 32), on pages 72-73.
I would be most interested in learning more about this phenomenon, in other references to it in the literature, and in corresponding with others who are interested in it.
-----
30th Nov 2011 01:37 UTCReiner Mielke Expert
30th Nov 2011 01:44 UTCRock Currier Expert




Mindat.org is an outreach project of the Hudson Institute of Mineralogy, a 501(c)(3) not-for-profit organization.
Copyright © mindat.org and the Hudson Institute of Mineralogy 1993-2024, except where stated. Most political location boundaries are © OpenStreetMap contributors. Mindat.org relies on the contributions of thousands of members and supporters. Founded in 2000 by Jolyon Ralph.
Privacy Policy - Terms & Conditions - Contact Us / DMCA issues - Report a bug/vulnerability Current server date and time: April 19, 2024 11:22:58
Copyright © mindat.org and the Hudson Institute of Mineralogy 1993-2024, except where stated. Most political location boundaries are © OpenStreetMap contributors. Mindat.org relies on the contributions of thousands of members and supporters. Founded in 2000 by Jolyon Ralph.
Privacy Policy - Terms & Conditions - Contact Us / DMCA issues - Report a bug/vulnerability Current server date and time: April 19, 2024 11:22:58