Axel Emmermann's Photo Gallery
LPK-NRKCalcite CaCO3 , Caliche
Nuevo León, Francisco I. Madero Municipality, Coahuila, MexicoDimensions: 120 mm x 75 mm x 45 mm
Field of View: 16.0 cm
Violet fluorescing calcite with crusts of orange fluorescing "caliche". The green fluorescence is caused by unknown organic matter and has a strong afterglow.
Mindat.org Photo of the Day - 10th Sep 2022
85L-VG2Scheelite Ca(WO4) , Calcite CaCO3
Dongshan Mine, Xianghualing Sn-polymetallic ore field, Linwu Co., Chenzhou, Hunan, ChinaDimensions: 110 mm x 70 mm x 35 mm
Field of View: 50 mm
A scheelite crystal sits next to a calcite crystal on a matrix of nonfluorescent fluorite. This photo was taken in UV-C and UV-B (254 nm and 312 nm) combined.
QMD-UWEWillemite Zn2SiO4 , Calcite CaCO3 , Tephroite Mn2+2SiO4
Sterling Mine, Sterling Hill, Ogdensburg, Sussex County, New Jersey, USADimensions: 70 mm x 55 mm x 32 mm
Red fluorescent calcite and green fluorescent willemite in non-fluorescent tephroite. The photo was taken under short wave UV (254 nm). Vey peculiar are the thin parallel lines of green fluorescent willemite. These were formed due to exsolution of the willemite from the original zincoan tephroite. When that originally very hot Zn-tephroite body cooled over a considerable period of time, the zinc ions no longer fitted in the tephroite structure and were squeezed out as the zinc slicate: willemite. This willemite formed in the parallel cracks, which formed due to the shrinking of the cooling rock. Logically, the cracks formed along the crystal planes of the tephroite. Utimately, this is the reason why these thin fluorescent lines are so neatly ordered. Both calcite and willemite fluoresce due to the presence of divalent manganese, which was derived from the surrounding tephroite, a manganese silicate. One would expect the tephroite to fluoresce too as it is , after all, a silicate of manganese, the ion that causes the fluorescence in calcite and willemite. It is, however, non-fluorescent due to a phenomenon called concentration-quenching.
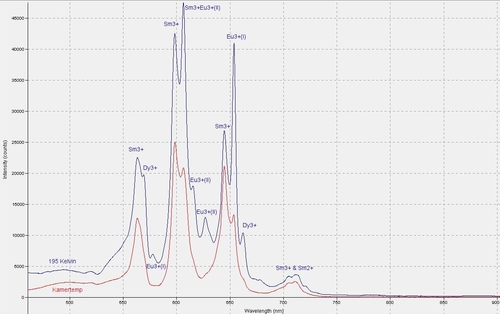
Fluorescence spectra of apatite from Jumilla, Spain under 405 nm laser
The spectra of this apatite from Jumilla, Spain, demonstrate the presence of quite a few Rare Earth Elements. There are noticeable differences between the spectrum measured at room temperature and the one at 195 Kelvin (dry ice). Measured with an Ocean Optics Jaz spectrometer of the Mineralogical Society of Antwerp.6CR-MM6Apatite Ca5(PO4)3(Cl/F/OH)
Jumilla, Murcia, SpainThe specimen's height is approximately 4 cm.
The orange-pink fluorescing apatite is embedded in "jumillaite", a mixture of calcite, analcime, leucite and other hydrothermally formed minerals in the volcanic rock of Jumilla, Murcia, Spain. Jumillaite often fluoresces white to sky-blue and has a strong phosporescence in the same color.
Photo taken with a Canon EOS 400D with NIKKOR 55mm Macro lens: f11, 6 seconds, ISO 400. UV-source (253.7 nm) is a UV-Systems SuperBright SW 2000.