Home PageAbout MindatThe Mindat ManualHistory of MindatCopyright StatusWho We AreContact UsAdvertise on Mindat
Donate to MindatCorporate SponsorshipSponsor a PageSponsored PagesMindat AdvertisersAdvertise on Mindat
Learning CenterWhat is a mineral?The most common minerals on earthInformation for EducatorsMindat ArticlesThe ElementsThe Rock H. Currier Digital LibraryGeologic Time
Minerals by PropertiesMinerals by ChemistryAdvanced Locality SearchRandom MineralRandom LocalitySearch by minIDLocalities Near MeSearch ArticlesSearch GlossaryMore Search Options
The Mindat ManualAdd a New PhotoRate PhotosLocality Edit ReportCoordinate Completion ReportAdd Glossary Item
Mining CompaniesStatisticsUsersMineral MuseumsClubs & OrganizationsMineral Shows & EventsThe Mindat DirectoryDevice SettingsThe Mineral Quiz
Photo SearchPhoto GalleriesSearch by ColorNew Photos TodayNew Photos YesterdayMembers' Photo GalleriesPast Photo of the Day GalleryPhotography
╳Discussions
💬 Home🔎 Search📅 LatestGroups
EducationOpen discussion area.Fakes & FraudsOpen discussion area.Field CollectingOpen discussion area.FossilsOpen discussion area.Gems and GemologyOpen discussion area.GeneralOpen discussion area.How to ContributeOpen discussion area.Identity HelpOpen discussion area.Improving Mindat.orgOpen discussion area.LocalitiesOpen discussion area.Lost and Stolen SpecimensOpen discussion area.MarketplaceOpen discussion area.MeteoritesOpen discussion area.Mindat ProductsOpen discussion area.Mineral ExchangesOpen discussion area.Mineral PhotographyOpen discussion area.Mineral ShowsOpen discussion area.Mineralogical ClassificationOpen discussion area.Mineralogy CourseOpen discussion area.MineralsOpen discussion area.Minerals and MuseumsOpen discussion area.PhotosOpen discussion area.Techniques for CollectorsOpen discussion area.The Rock H. Currier Digital LibraryOpen discussion area.UV MineralsOpen discussion area.Recent Images in Discussions
Mineral PhotographyUsable Magnification
17th Mar 2017 00:14 UTCRonald J. Pellar Expert
Maximum Usable Magnification
When using a microscope objective or a separate reversed lens there is a maximum useful magnification that you can get. Pushing the magnification beyond this point will only increase blur size (comparable to pixilation). The maximum useful magnification is computed by dividing the size of the camera sensor pixel size by the resolved dimension of the lens.
The resolved dimension of the lens can be computed from the diffraction limited formula:
r = 1.22ƛ(f/no.) = 0.611ƛ/(N.A.) [Numerical Aperture, N.A., is 1/2(f no.)]
where r, the resolved dimension, and ƛ, the wavelength, are in microns.
EXAMPLE: For a 35 mm APS-C 12 megapixel sensor the pixel size is 5.65 microns in my Nikon D300. With a f/2.8 lens (N.A. = 0.18) and ƛ = 0.5 micron (green) the resolved dimension is 1.71 microns. This gives a maximum usable magnification of 3.32x. Larger magnifications than this will simply enlarge the blur circle and fuzz up the image. NOTE: Higher megapixel sensors reduce the useful magnification further, e.g., a 25 megapixel APS-C sensor will have a maximum useful magnification of about 2.2x. A F format sensor of the same number of megapixels will increase the useful magnification by a factor of 1.5, e.g., the 35mm SLR, 12 megapixel F format sensor has a usable magnification of 4.8x.
To achieve larger useful magnification, a second lens is required to increase the apparent size of the camera pixel (one of the reasons that a microscope has an eyepiece). One way to do this is to use a wide-angle lens on the camera along with the microscope objective (an infinity focus objective makes it easier to couple the microscope objective to the camera). The short focal length (wide-angle) lens will essentially magnify the pixel size in the camera to increase the maximum usable magnification. This will be especially useful for the high megapixel cameras, i.e., > 12 megapixels, where the camera pixel size is smaller.
An example of this problem is shown here:
where I used a Nikon D300 (12 megapixel APS-C sensor) with 7x objective on a Mikon trinocular microscope and the image appears slightly fuzzy due to pushing the magnification above the maximum 3.32x. Compare this image with this one:
where I used the same setup as above but with the 4x objective. Both images have the same pixel density for comparison purposes.
Visual Magnification
The maximum usable magnification, referred to above, is with reference to the camera sensor plane. For viewing purposes on some sort of display, the total usable magnification occurs when;
1) A pixel in the image being viewed contains information from one pixel in the camera sensor.
2) The person viewing the image is at an optimum distance from the image, i.e., the person viewing the image can just barely resolve a single pixel.
EXAMPLE: If I view an image in Photoshop at 100% on my HD monitor that is 20” wide, a single pixel is 257 microns in size and the actual magnification is 45.5x relative to the camera. If the camera was setup to provide an image at the maximum usable magnification as described above, the total magnification from the object to the monitor is 151x. If the viewer is further away than the optimal distance for viewing (violating the second criteria above), then the magnification can be increased, either optically or electronically, e.g., in Photoshop setting the image size greater then 100%. NOTE: increasing the magnification electronically beyond 100% can cause pixilation if the software does not use interpolation. With interpolation the image just gets fuzzier.
As the camera pixel size changes, e.g., switching from a 12 megapixel camera to a 25 megapixel camera with the same sensor format, e.g., APS-C,, choosing and objective magnification to maintain maximum usable magnification does not change the visual magnification! The maximum usable visual magnification will always be the viewed image pixel size divided by the objective lens resolved dimension, e.g., a 257 micron image pixel divided by 1.7 micron resolved dimension from the previous example gives a total magnification of 151x. In other words, all intervening factors like camera sensor size, secondary lenses, electronic magnification, etc. cannot improve the visual usable magnification. They can only make things worse not better.
Using Magnification Below Maximum
When the camera setup is such that a single camera pixel size does is greater than the lens resolved dimension, the image will be subsampled, i.e., there is information incident on the pixel that is averaged over the sensor pixel. This subsampling can cause aliasing error and/or moiré. Most digital SLR cameras having less than 36 megapixels use an antialiasing optical filter to alleviate this problem. When the camera pixel is equivalent to the resolved dimension of the lens, the need for an antialiasing filter goes away. This condition satisfies the Nyquist or Shannon condition from information theory and should be considered the optimal camera setup as this condition retains sharpness and limits aliasing errors.
Microscope Objective vs. Reversing Lens
The discussion and calculations used above assume that the lens being used to image the object (objective lens) is diffraction limited and that higher magnifications are being used than 1:1. At magnifications close to 1:1, the diffraction limit formula becomes;
r = 1.22ƛLo/D
where Lo is the distance from the center of the lens to the object being imaged. As the magnification becomes higher Lo approaches the focal length of the lens thereby approaching the diffraction limited formula. In general this object distance is greater then the lens focal length and the resolved dimension increases slightly, e.g., a maximum of a factor of 2 at a magnification 1:1. At less than 1:1 magnification the Lo term becomes Li, the image distance.
Another factor is the lens aberrations that can be greater than the diffraction limit at small f/no. The f/no that is optimal is where the aberration blur equals the diffraction blur and usually referred to as the “sweet spot”. This sweet spot is usually around 2x the minimum f/no of the lens, e.g., a f/2.8 lens will usually have its sweet spot at f/5.6 to f/8. Some lenses are notorious for the excellance of their resolving power even at maximum aperature, e.g., the older Micro-Nikkor 55mm macro lens. The lenses are designed to correct the aberrations at the focal plane where the image size is smaller than the object size. In the case of macrophotography, i.e., magnification greater than 1:1, it makes sense to reverse the lens and place the object in the near field.
Microscope objective lenses are specially designed for minimizing the resolution dimension at full aperature. Since the aperture is not adjustable, f/no does not have the same usefulness as for camera lenses and they are defined by their numerical aperture (defined above with respect to f/no). The higher the N. A. the better resolving power and much effort is put into achieving the maximum resolving power thereby increasing their price considerably. Adding more correction for color dispersion in the lens materials can raise the price even more, e.g., apochromats cost more than achromats.
Finite focus objective lenses are usually designated by a magnification power to facilitate the calculation of overall magnification with an eyepiece. This magnification is the ration between the image distance and the object distance relative to that lens. An eyepiece magnifies the real image to be viewed. However, a camera sensor can be placed in the image plane directly which can limit the usable magnification. Another lens can be used to magnify the camera sensor pixels for use with higher magnification objective lenses.
Infinite focus objective lenses are designed specifically for use with another lens to focus the image. The second lens can be an eyepiece or a camera lens.
Conclusion
The micro/macro setup of lens and viewing image should be such that a pixel in the viewed image corresponded to the resolved dimension in the objective lens.
MUMs (Maximum Usable Magnification) the word!
17th Mar 2017 00:19 UTCRob Woodside 🌟 Manager

3rd Dec 2017 18:15 UTCLarry Eicher
Thanks for presenting these thoughts. After reading this, I found a used Canon EOS xsi rebel. I used it for the longest time making images. At one point this fall I came to the conclusion I didn't need to use it and went back to my Canon 7D, 18 megapixel which I prefer using. I must admit that I am confused in trying to observe what you describe. I look at that math and feel incapable of making the calculations. I wonder if anyone could help me.
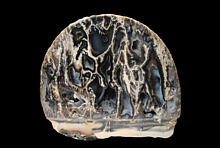
I recently made an image of Clinoptilolite from Los Angeles County California. I used the rebel above, I mounted my Sigma 105mm lens on 68mm of extension and a 1.4x teleconverter. The closeup filter was a Yashica 28mm cine lens reversed. Below is the result. I used 1/20 second shutter and f/5.6 aperture.
I am still confused when I read your above essay. I can see that there is less sharpness in this than in other images I make with less Magnification. This image has a FOV 2mm and is 11.75 on the sensor. What do you think? Did I exceed my MUM in making this?
Confusedly Yours,
Larry Eicher
4th Dec 2017 23:18 UTCRonald J. Pellar Expert
You have a rather complicated setup. For the purposes of finding the Maximum Usable Magnification (MUM) you only need to consider the one lens focused upon th specimen. All other lenses, extensions, etc. only serve to magnify the sensor element in the camera.
I will assume that the 28mm Cine lens reversed is used with specimen at the focal plane position. The photo was at f/5.6 so assuming that the blur circle is diffraction limited (close for the f/no. at the sweet spot0 of approx.. 2x full aperture) the resolving power at the specimen is about 3 microns, from the formula for diffraction limit in my previous post
. If the field of view is 2mm than the MUM is reached if the number of pixels in the final image from the camera has 2000 microns divided by 3 microns equals 667 pixels. Anymore pixels than that and your image will look blurred at 100% viewing size, i.e., 1 pixel in the image equals 1 or more pixels on the computer monitor.
Any further degradation in resolving power in the subsequent setup will only increase the minimum resolved size thereby reducing the usable number of pixels to achieve the MUM condition. Simplifying the setup to the imaging lens and a camera sensor by means of a bellows would reduce the number possible sources for degradation.
Not knowing what the number of pixels are in your image, I am unable to judge where your are regarding the MUM point. But I suspect that you may have exceeded it.
To Summarize:
1) Compute the best resolved size with lens that you are using to focus on the specimen using the "sweet spot" f/no. and the diffraction limit formula, in microns.
2) Determine the FOV of the desired image in mm and convert to microns.
3) Divide the FOV in microns by the resolving power in microns and you will have the MUM condition for setup in Pixels.
4) Adjust the magnification in your setup to give you number of MUM number of pixels just calculated, to match number of pixels in the corresponding direction on your camera senor.
If you calculate that you have more pixels across the image than the MUM criteria, then you can lower the magnification and get more of the specimen into the image at the same resolving power.
If you have fewer pixels across the image then you can increase the magnification, i.e., reduce the FOV, and get optimum resolving power (the MUM condition).
5th Dec 2017 00:14 UTCJolyon Ralph Founder
So if you have a 50 megapixel sensor (8688 x 5792 pixels) but you want to create a final image that is 2172 x 1448 pixels (ie 1/4 on both dimensions) to end up with a 3 megapixel image then you only need to worry about the dot pitch on your sensor as if it was a 3 megapixel sensor.
Of course this will also give you the advantage of downsampling to get greater sensitivity than you would on a simple 3 megapixel sensor - plus it counters any artifacts that the bayer filter may have introduced.
5th Dec 2017 18:12 UTCRonald J. Pellar Expert
Your point is for down sampled images. Cropping to the new size has no effect.
5th Dec 2017 20:51 UTCVolker Betz 🌟 Expert
I am a litte confused about this discussion. This may be due to my limitations in english.
I have a more parctical approach. The limiting factor is the resolution of the lens.
With plan apochromatic lenses (20 x or 40x) a resolution of ~1 µm is possible.
This is theory as vibrations are a limiting factor. With 20 x enlargement the resolution ist 20 µm on the sensor.
Under this conditions the most cameras fullfil the MFT critera of 4 pixel per line pair. Micro 4/3 cameras do better because their pixels are smaller.
If we use a 15 mm (15000 µm) sensor hight and devide that by a 20 µm resolution then we get 750 sharp points per sensor hight.
This will be recognised as "sharp" by the human eye. HD TV has 1080 pixels per screen hight.
So to my opinion 20 x works well, some even use 40 X . This means that fields of view of about 0.8 mm in a picture of minerals is possible. But ist is of course better for the picture, if individual crystals are not smaller than ~300 µm.
This 300 µm per crystal condition is about the practical limit after my 12 years of stacking.
Not to forget that proper lightnig and post procesing has a big influence on the "perormance" of a picture.
And less details make a picture better.
Volker
5th Dec 2017 21:24 UTCJolyon Ralph Founder
Yes, I should have been clearer, that's for downsampling the full image, not cropping!

8th Dec 2017 15:32 UTCLarry Eicher
When you say, "you only need to consider the one lens focused on the specimen," do you mean the point at which it in focus? I use the 105 focused on it's closest setting. I then fine-tune focusing with Adorama Focusing Rail moving the assembly closer or further to achieve my starting point for the stack.
What does, "with specimen at the focal plane position." mean? Does it mean the point at which it is in focus?
The pixel dimensions of the sensor on the Rebel XSI is 4272 by 2848. I am trying to process
the final image to those dimensions. I have started using PS CC2017 and don't have it under control
as yet. It changes things and doesn't tell me. I have gone back and checked my procedure for the
image I shared. I found one thing it pulled on me. I am processing it again. This time the number
of pixels on the sensor are equal to the number in the image.
What if I were to set my 7D to capture at those dimensions?
Why is a bellows so much better than extension tubes? I have two sets each equaling 68mm. I can't
imagine a bellows, at least those I have seen, extending the lens further than that. Is it because
it is infinitely variable between the extremes. I don't see bellows for sale listing the amount
of extension available. I seem to understand that your suggestion is for me to simplify my setup.
I have made images using only extension tubes and the 105mm. I have tried using two sets of
extension tubes (136mm). I don't think I was recording the FOV and X factor on the sensor back than. The images attached was created using a sigma 105mm mounted on 136mm of extension, (no filter and no 1.4X tele converter). The series contains 45 captures and was shot on an 18mp 7D.
I have looked into microscope objectives. In fact I own a microscope from which I could steal
objectives. I think that I would need objectives with an aperture adjustment built in unless
I mounted it on my 105mm.
My sensor measures 23,500 microns on the long side. I don't know how to relate this to the wave
length. Don't all colors have different wave lengths? Do you average the wave lengths of the
colors RGB?
FYI, the other attached images is supposed to be the new and improved version. Can you see a difference? I think I can.
Regards,
Larry Eicher
9th Dec 2017 21:56 UTCRonald J. Pellar Expert
The most important lens, i.e., primary lens, is the lens that collects the light directly from the specimen. That lens contains the aberration’s (iris, f/no setting) that causes the diffraction along with all the lens designers corrections for aberrations in the lens. All subsequent lenses, e.g., relay lenses, lens on the camera body if its not the primary lens, etc., can only make matters worse but not better. The diffraction blurring and aberration blurring effects occur whether the lens is in focus or not. However, our concern is for the quality of the image at the in focus pixels which are determined by focusing the primary lens by whatever means you choose to adjust the focus point.
At a magnification of 1:1, or 100%, the distance to the focal plane is twice the focal length of the primary lens. The macro lens designer does his best to reduce the aberrations within the image out to this image distance so that lens performance is maintained over this range, i.e., from the focal length to 2x the focal length. At higher magnifications than 1:1 lens corrections are not as good. To avoid this degradation at higher magnifications you can reverse the lens so that all corrections that the lens designer put in are again effective. Using extension tubes or bellows, require a reversing adapter so the object being imaged is on the side of the lens that the sensor would be normally.
The question of whether to use an extension tube or a bellows is a bit complex. Most macrophotography utilize a tripod. Extension tubes between the camera body and the lens change the center of gravity further away from the camera body mounted on the tripod and create a cantilever situation that can increase the effects of vibration. Most bellows mounts have a tripod adapter that places the C.G. of setup over the tripod and should be less sensitive to vibrations. In addition, some bellows allow for parallax/perspective adjustments that are not possible with extension tubes. Using multiple extension tubes in a stacked configuration can be slightly looser due to tolerances in the mounts and this could add to vibration sensitivity. The bellows provides more flexibility in adjusting the magnification ratio desired as extension tubes are fixed in length. There are many bellows available with up to more than 200mm extension.
The “focal plane” is an imaginary plane on the imaging side of the primary lens, as opposed to the object or specimen side of the lens, at a fixed distance from the primary lens over which anything in that plane is in focus. If the primary lens is set at infinity focus, the focal plane distance for the lens is known as the ‘focal length”. For macrophotography, the distance to the focal plane is greater than the focal length and at a magnification of 1:1 the focal plane is twice the focal length of the lens.
The pixel size within the camera can be calculated by dividing the largest number of pixels into the largest dimension of the camera sensor, e.g., an APS-C sensor has a maximum dimension of 24mm and if your sensor maximum number of pixels is 4272 the pixel size is 24,000/4,272 = 5.62 microns. If the primary lens is the only lens in the macro setup then the MUM condition can be determined by dividing the pixel size by the resolving power of the primary lens, e.g., for a diffraction limited blur circle for a setting of f/5.8 the resolving power is about 2 microns and MUM condition is a magnification of slightly less than 3x.
The diffraction blur is calculated from the formula give previously using a wavelength of 510nm or 0.51 microns. This wavelength is close to the middle of the visual wavelength range but more importantly it is near the peak for maximum acuity in the human eye. Our eyes are not ver good at focusing blue light and are only slight better at focusing red light so using an wavelength value in the green gives pretty goo results.
Another way to see if your image is at the MUM condition is to view it in imaging software at 100% size. In Photoshop or Lightroom the image as opened will usually be at some percentage less than 100% if the whole image is to be visible. This percentage refers to the ration between a pixel size in the image to the pixel size on your monitor. This percentage can be found in title to the image in PS. In PS the magnification can be changed using the “Ctl +” or “Ctl –“ keys. As you approach 100% the image should look sharp. At the MUM condition the image will be sharp at the 100% point as well. At 200% you will notice blurring. If blurring is noticeable prior to reaching the 100% point then you have exceeded the MUM condition.
Most macro lenses are limited to an f/no of f/2.8 or higher, this limits the resolving power to about 2-3 microns at best. Better information on a given lens can usually be found at the lens manufacturer’s website as a plot of resolving power in line pairs per mm versus f/no. These plots give two important pieced of info for macrophotography, the f/no that corresponds to the “sweet spot” and the number of line pairs per mm at that spot. The resolving power in microns can be found by dividing 500 by the maximum line pairs per millimeter, i.e., a millimeter is 1000 microns, a line pair is one white line followed by one black line so the resolving power is i/2 a line pair.
Microscope objectives are specially designed for high magnification and great pains are taken to correct the aberrations and much lower f/no are achieved. An f/1.0 can be found which can give a resolving power less than 1 micron. However, microscope objectives are rated not by f/no but by Numerical Aperture, N.A., which relates to f/no by NA = 1/2fno, e.g., a NA of 0.5 is the same as f/1.0. Since resolving power is so important for microscope objectives the best ones do more correction of color aberrations than an achromatic lens. These specially corrected lens are referred to as “apochromats”. Of course these objectives can get expensive, deservedly so. For photomicrography/macrophotography a microscope objective with NA greater than 0.1 will yield better resolving power than macro camera lenses as a NA of 0.1 corresponds to an f/5 which is about what the sweet spot for a macro lens is at. The microscope objective, that is not an infinity focus type, should be used with a bellows a camera body only. This will provide better adjustments for magnification without introducing other lens that if not properly setup could degrade performance. Microscope objectives that are designated infinity focus require a second lens to refocus the image on the camera sensor thus complicating the setup. Do not use microscope objectives designed for use with immersion oil as they will not perform well in air.




Mindat.org is an outreach project of the Hudson Institute of Mineralogy, a 501(c)(3) not-for-profit organization.
Copyright © mindat.org and the Hudson Institute of Mineralogy 1993-2024, except where stated. Most political location boundaries are © OpenStreetMap contributors. Mindat.org relies on the contributions of thousands of members and supporters. Founded in 2000 by Jolyon Ralph.
Privacy Policy - Terms & Conditions - Contact Us / DMCA issues - Report a bug/vulnerability Current server date and time: April 25, 2024 05:31:08
Copyright © mindat.org and the Hudson Institute of Mineralogy 1993-2024, except where stated. Most political location boundaries are © OpenStreetMap contributors. Mindat.org relies on the contributions of thousands of members and supporters. Founded in 2000 by Jolyon Ralph.
Privacy Policy - Terms & Conditions - Contact Us / DMCA issues - Report a bug/vulnerability Current server date and time: April 25, 2024 05:31:08