Quenstedtite
A valid IMA mineral species - grandfathered
This page is currently not sponsored. Click here to sponsor this page.
About Quenstedtite
Formula:
Fe2(SO4)3 · 11H2O
Colour:
White to very pale violet to red-violet; colorless to pale rose in transmitted light.
Lustre:
Sub-Vitreous, Silky
Hardness:
2½
Specific Gravity:
2.11 - 2.15
Crystal System:
Triclinic
Name:
Named in 1888 by Gottlob Eduard Linck in honor of Friedrich August von Quenstedt [July 10, 1809 Eisleben, Saxony, Germany – December 21, 1889], professor of mineralogy and geology at Eberhard Karls Universität in Tübingen. Quenstedt was also noted for his paleontological research (ammonites and pterodactyls) and a fossil turtle, Proganochelys quenstedti, is named for him as well as five genera of other extinct species (2 coenlenterates, 1 ammonite, 1 mollusc, and 1 crinoid).
Type Locality:
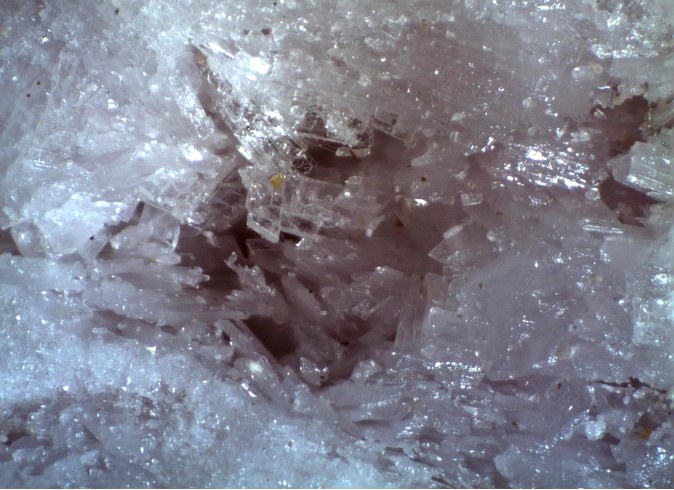
Straight fibrous to columnar crystals with a bright luster and fibrous appearance
Unique Identifiers
Mindat ID:
3341
Long-form identifier:
mindat:1:1:3341:7
GUID
(UUID V4):
(UUID V4):
5f7ae427-0d96-4bf1-b761-c9ffbb84be3f
IMA Classification of Quenstedtite
Approved, 'Grandfathered' (first described prior to 1959)
IMA Formula:
Fe3+2(SO4)3 · 11H2O
First published:
1888
Classification of Quenstedtite
7.CB.65
7 : SULFATES (selenates, tellurates, chromates, molybdates, wolframates)
C : Sulfates (selenates, etc.) without additional anions, with H2O
B : With only medium-sized cations
7 : SULFATES (selenates, tellurates, chromates, molybdates, wolframates)
C : Sulfates (selenates, etc.) without additional anions, with H2O
B : With only medium-sized cations
29.8.5.1
29 : HYDRATED ACID AND NORMAL SULFATES
8 : A2(XO4)3·H2O
29 : HYDRATED ACID AND NORMAL SULFATES
8 : A2(XO4)3·H2O
25.10.11
25 : Sulphates
10 : Sulphates of Fe alone
25 : Sulphates
10 : Sulphates of Fe alone
Mineral Symbols
As of 2021 there are now IMA–CNMNC approved mineral symbols (abbreviations) for each mineral species, useful for tables and diagrams.
| Symbol | Source | Reference |
|---|---|---|
| Qst | IMA–CNMNC | Warr, L.N. (2021). IMA–CNMNC approved mineral symbols. Mineralogical Magazine, 85(3), 291-320. doi:10.1180/mgm.2021.43 |
Physical Properties of Quenstedtite
Sub-Vitreous, Silky
Transparency:
Transparent, Translucent
Colour:
White to very pale violet to red-violet; colorless to pale rose in transmitted light.
Streak:
White
Hardness:
2½ on Mohs scale
Tenacity:
Brittle
Cleavage:
Perfect
Perfect on {010}
Good on {100}
Perfect on {010}
Good on {100}
Fracture:
Fibrous
Density:
2.11 - 2.15 g/cm3 (Measured) 2.14 g/cm3 (Calculated)
Optical Data of Quenstedtite
Type:
Biaxial (+)
RI values:
nα = 1.547 nβ = 1.566 nγ = 1.594
2V:
Measured: 70° , Calculated: 82°
Max Birefringence:
δ = 0.047

Image shows birefringence interference colour range (at 30µm thickness)
and does not take into account mineral colouration.
and does not take into account mineral colouration.
Surface Relief:
Low
Dispersion:
Strong r
Optical Extinction:
X(–43°,45°); Y(128°,43°); Z(–138°,88°) with c(0°,0°) and b*(0°,90°) using (φ,ρ).
Chemistry of Quenstedtite
Mindat Formula:
Fe2(SO4)3 · 11H2O
Elements listed:
Crystallography of Quenstedtite
Crystal System:
Triclinic
Class (H-M):
1 - Pinacoidal
Space Group:
P1
Cell Parameters:
a = 6.184(5) Å, b = 23.6(2) Å, c = 6.539(5) Å
α = 94.18(8)°, β = 101.73(8)°, γ = 96.27(8)°
α = 94.18(8)°, β = 101.73(8)°, γ = 96.27(8)°
Ratio:
a:b:c = 0.262 : 1 : 0.277
Unit Cell V:
924.35 ų (Calculated from Unit Cell)
Z:
2
Morphology:
Aggregates of minute crystals, tabular {010}, or short prismatic [100], highly modified at times.
Twinning:
Common on {010}.
Crystal Structure
Load
Unit Cell | Unit Cell Packed
2x2x2 | 3x3x3 | 4x4x4
Unit Cell | Unit Cell Packed
2x2x2 | 3x3x3 | 4x4x4
Show
Big Balls | Small Balls | Just Balls | Spacefill
Polyhedra Off | Si Polyhedra | All Polyhedra
Remove metal-metal sticks
Big Balls | Small Balls | Just Balls | Spacefill
Polyhedra Off | Si Polyhedra | All Polyhedra
Remove metal-metal sticks
Display Options
Black Background | White Background
Perspective On | Perspective Off
2D | Stereo | Red-Blue | Red-Cyan
Black Background | White Background
Perspective On | Perspective Off
2D | Stereo | Red-Blue | Red-Cyan
View
CIF File Best | x | y | z | a | b | c
CIF File Best | x | y | z | a | b | c
Rotation
Stop | Start
Stop | Start
Labels
Console Off | On | Grey | Yellow
Console Off | On | Grey | Yellow
Data courtesy of the American Mineralogist Crystal Structure Database. Click on an AMCSD ID to view structure
| ID | Species | Reference | Link | Year | Locality | Pressure (GPa) | Temp (K) |
|---|---|---|---|---|---|---|---|
| 0000409 | Quenstedtite | Thomas J N, Robinson P D, Fang J H (1974) Crystal structures and mineral chemistry of hydrated ferric sulfates. IV. The crystal structure of quenstedtite American Mineralogist 59 582-586 |  | 1974 | 0 | 293 |
CIF Raw Data - click here to close
X-Ray Powder Diffraction
Powder Diffraction Data:
| d-spacing | Intensity |
|---|---|
| 4.08 Å | (very very strong) |
| 5.78 Å | (very strong) |
| 4.19 Å | (very strong) |
| 3.80 Å | (strong) |
| 5.03 Å | (moderately strong) |
| 5.57 Å | (moderately weak) |
| 5.34 Å | (moderately weak) |
Comments:
Tierra Amarilla, Chile. The data are from Cesbron (1964).
Geological Environment
Paragenetic Mode(s):
| Paragenetic Mode | Earliest Age (Ga) |
|---|---|
| Stage 7: Great Oxidation Event | <2.4 |
| 47a : [Near-surface hydration of prior minerals] | |
| 47b : [Sulfates and sulfites] |
Geological Setting:
Oxidized zone of pyrite rich orebodies, especially in arid climates.
Type Occurrence of Quenstedtite
General Appearance of Type Material:
Small reddish-violet gypsum-like tablets up to 5 mm long, 1-2 mm wide and 1 mm thick.
Place of Conservation of Type Material:
Muséum Nationale d’Histoire Naturelle, Paris, France.
Geological Setting of Type Material:
Sulfate deposit. Oxidized zone of a pyrite-rich deposit in a desert.
Other Language Names for Quenstedtite
Common Associates
Associated Minerals Based on Photo Data:
Related Minerals - Strunz-mindat Grouping
| 7.CB. | Sarvodaite | Al2(SO4)3 · 5H2O |
| 7.CB.02 | Voudourisite | CdSO4 · H2O |
| 7.CB.05 | Dwornikite | Ni(SO4) · H2O |
| 7.CB.05 | Gunningite | ZnSO4 · H2O |
| 7.CB.05 | Kieserite | MgSO4 · H2O |
| 7.CB.05 | Poitevinite | (Cu,Fe)SO4 · H2O |
| 7.CB.05 | Szmikite | MnSO4 · H2O |
| 7.CB.05 | Szomolnokite | FeSO4 · H2O |
| 7.CB.05 | Cobaltkieserite | CoSO4 · H2O |
| 7.CB.07 | Sanderite | MgSO4 · 2H2O |
| 7.CB.10 | Bonattite | CuSO4 · 3H2O |
| 7.CB.12 | Belogubite | CuZn(SO4)2 · 10H2O |
| 7.CB.15 | Aplowite | (Co,Mn,Ni)SO4 · 4H2O |
| 7.CB.15 | Boyleite | (Zn,Mg)SO4 · 4H2O |
| 7.CB.15 | Ilesite | (Mn,Zn,Fe)SO4 · 4H2O |
| 7.CB.15 | Rozenite | FeSO4 · 4H2O |
| 7.CB.15 | Starkeyite | MgSO4 · 4H2O |
| 7.CB.15 | Drobecite | CdSO4 · 4H2O |
| 7.CB.15 | Cranswickite | MgSO4 · 4H2O |
| 7.CB.20 | Chalcanthite | CuSO4 · 5H2O |
| 7.CB.20 | Jôkokuite | MnSO4 · 5H2O |
| 7.CB.20 | Pentahydrite | MgSO4 · 5H2O |
| 7.CB.20 | Siderotil | FeSO4 · 5H2O |
| 7.CB.25 | Bianchite | Zn(SO4) · 6H2O |
| 7.CB.25 | Chvaleticeite | Mn(SO4) · 6H2O |
| 7.CB.25 | Ferrohexahydrite | FeSO4 · 6H2O |
| 7.CB.25 | Hexahydrite | MgSO4 · 6H2O |
| 7.CB.25 | Moorhouseite | Co(SO4) · 6H2O |
| 7.CB.25 | Nickelhexahydrite | Ni(SO4) · 6H2O |
| 7.CB.30 | Retgersite | NiSO4 · 6H2O |
| 7.CB.35 | Bieberite | CoSO4 · 7H2O |
| 7.CB.35 | Boothite | CuSO4 · 7H2O |
| 7.CB.35 | Mallardite | MnSO4 · 7H2O |
| 7.CB.35 | Melanterite | Fe2+(H2O)6SO4 · H2O |
| 7.CB.35 | Zincmelanterite | (Zn,Cu,Fe)SO4 · 7H2O |
| 7.CB.35 | Alpersite | (Mg,Cu)(SO4) · 7H2O |
| 7.CB.40 | Epsomite | MgSO4 · 7H2O |
| 7.CB.40 | Goslarite | ZnSO4 · 7H2O |
| 7.CB.40 | Morenosite | NiSO4 · 7H2O |
| 7.CB.45 | Alunogen | Al2(SO4)3 · 17H2O |
| 7.CB.45 | Meta-alunogen | Al2(SO4)3 · 12H2O |
| 7.CB.50 | Aluminocoquimbite | Al2Fe2(SO4)6(H2O)12 · 6H2O |
| 7.CB.50 | Lazaridisite | 3CdSO4 · 8H2O |
| 7.CB.52 | Pararaisaite | CuMg[Te6+O4(OH)2] · 6H2O |
| 7.CB.55 | Coquimbite | AlFe3(SO4)6(H2O)12 · 6H2O |
| 7.CB.55 | Paracoquimbite | Fe4(SO4)6(H2O)12 · 6H2O |
| 7.CB.55 | Rhomboclase | (H5O2)Fe3+(SO4)2 · 2H2O |
| 7.CB.55 | Raisaite | CuMg[Te6+O4(OH)2] · 6H2O |
| 7.CB.57 | Caichengyunite | Fe2+3Al2(SO4)6 · 30H2O |
| 7.CB.60 | Kornelite | Fe2(SO4)3 · 7H2O |
| 7.CB.70 | Lausenite | Fe2(SO4)3 · 5H2O |
| 7.CB.75 | Lishizhenite | ZnFe2(SO4)4 · 14H2O |
| 7.CB.75 | Römerite | Fe2+Fe3+2(SO4)4 · 14H2O |
| 7.CB.80 | Ransomite | CuFe2(SO4)4 · 6H2O |
| 7.CB.85 | Apjohnite | Mn2+Al2(SO4)4 · 22H2O |
| 7.CB.85 | Bílinite | Fe2+Fe3+2(SO4)4 · 22H2O |
| 7.CB.85 | Dietrichite | (Zn,Fe2+,Mn2+)Al2(SO4)4 · 22H2O |
| 7.CB.85 | Halotrichite | FeAl2(SO4)4 · 22H2O |
| 7.CB.85 | Pickeringite | MgAl2(SO4)4 · 22H2O |
| 7.CB.85 | Redingtonite | (Fe2+,Mg,Ni)(Cr,Al)2(SO4)4 · 22H2O |
| 7.CB.85 | Wupatkiite | (Co,Mg,Ni)Al2(SO4)4 · 22H2O |
| 7.CB.90 | Meridianiite | MgSO4 · 11H2O |
Other Information
Notes:
Easily soluble in water.
Health Risks:
No information on health risks for this material has been entered into the database. You should always treat mineral specimens with care.
Internet Links for Quenstedtite
mindat.org URL:
https://www.mindat.org/min-3341.html
Please feel free to link to this page.
Please feel free to link to this page.
Search Engines:
External Links:
Mineral Dealers:
References for Quenstedtite
Reference List:
Ungemach, Henri (1935) Sur certains minéraux sulfatés du Chili. Bulletin de Minéralogie, 58 (3) 97-221 doi:10.3406/bulmi.1935.4369
Bandy, Mark C. (1938) Mineralogy of three sulphate deposits of northern Chile. American Mineralogist, 23 (11) 669-760
Localities for Quenstedtite
Locality List
 - This locality has map coordinates listed.
- This locality has map coordinates listed.
 - This locality has estimated coordinates.
ⓘ - Click for references and further information on this occurrence.
? - Indicates mineral may be doubtful at this locality.
- This locality has estimated coordinates.
ⓘ - Click for references and further information on this occurrence.
? - Indicates mineral may be doubtful at this locality.
 - Good crystals or important locality for species.
- Good crystals or important locality for species.
 - World class for species or very significant.
(TL) - Type Locality for a valid mineral species.
(FRL) - First Recorded Locality for everything else (eg varieties).
- World class for species or very significant.
(TL) - Type Locality for a valid mineral species.
(FRL) - First Recorded Locality for everything else (eg varieties).
All localities listed without proper references should be considered as questionable.
Argentina | |
| Morello et al. (2016) |
Canada | |
| Gomi (2001) |
Chile | |
| Färber (n.d.) |
| identified by Gerhard Möhn et al. (unlike Coquimbite) | |
| Kampf +5 other references |
| Natural History Museum Vienna collection (Uwe Kolitsch SXRD on Arturo Molina material) |
| Neues Jahrb.Min. (1888) +2 other references |
Germany | |
| Weiß (1990) |
| Wittern (2001) |
| Wittern (2001) |
| Der Aufschluss Vol.55 +2 other references |
Greece | |
| Schnorrer (1995) +1 other reference |
| Branko Rieck collection | |
Hungary | |
| Szakáll et al. (1996) |
| Hans van Zelfeld |
Japan | |
| Miura et al. (1994) |
Romania | |
| Ed.:Szakáll S.-Kristály ... |
Slovakia | |
| Anthony et al. (2016) |
Spain | |
| Calvo Rebollar et al. (2022) |
USA | |
| part 2 +5 other references |
| Yale Peabody Museum |
Quick NavTopAbout QuenstedtiteUnique IdentifiersIMA Classification Classification Mineral SymbolsPhysical Properties Optical Data Chemistry Crystallography Crystal StructureX-Ray Powder DiffractionGeological EnvironmentType Occurrence Other LanguagesCommon AssociatesStrunz-MindatOther InformationInternet Links References Localities Locality List





 symbol to view information about a locality.
The
symbol to view information about a locality.
The 



Slag dumps, Richelsdorf Smelter, Süß, Nentershausen, Hersfeld-Rotenburg, Kassel Region, Hesse, Germany