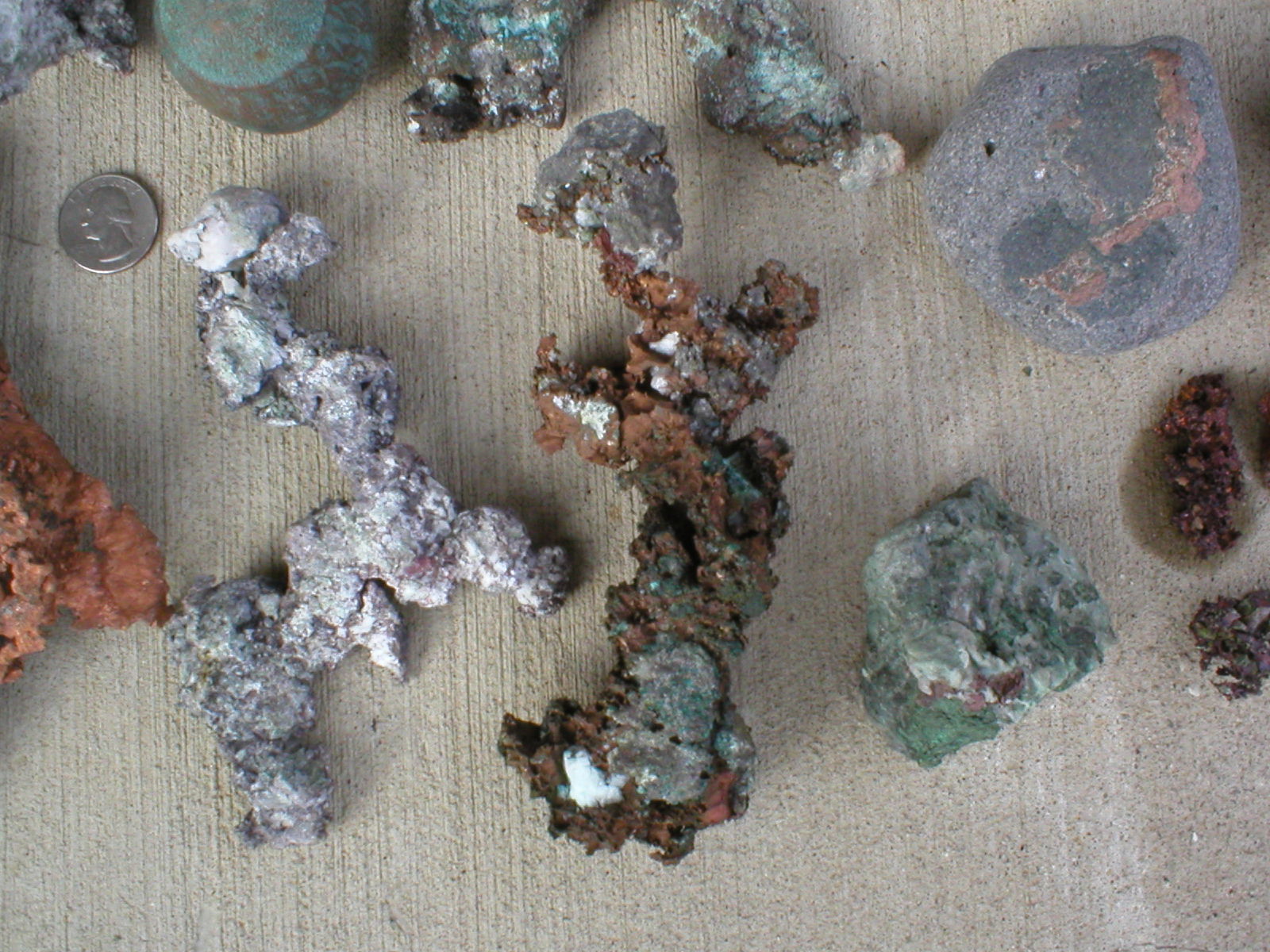Home PageAbout MindatThe Mindat ManualHistory of MindatCopyright StatusWho We AreContact UsAdvertise on Mindat
Donate to MindatCorporate SponsorshipSponsor a PageSponsored PagesMindat AdvertisersAdvertise on Mindat
Learning CenterWhat is a mineral?The most common minerals on earthInformation for EducatorsMindat ArticlesThe ElementsThe Rock H. Currier Digital LibraryGeologic Time
Minerals by PropertiesMinerals by ChemistryAdvanced Locality SearchRandom MineralRandom LocalitySearch by minIDLocalities Near MeSearch ArticlesSearch GlossaryMore Search Options
The Mindat ManualAdd a New PhotoRate PhotosLocality Edit ReportCoordinate Completion ReportAdd Glossary Item
Mining CompaniesStatisticsUsersMineral MuseumsClubs & OrganizationsMineral Shows & EventsThe Mindat DirectoryDevice SettingsThe Mineral Quiz
Photo SearchPhoto GalleriesSearch by ColorNew Photos TodayNew Photos YesterdayMembers' Photo GalleriesPast Photo of the Day GalleryPhotography
╳Discussions
💬 Home🔎 Search📅 LatestGroups
EducationOpen discussion area.Fakes & FraudsOpen discussion area.Field CollectingOpen discussion area.FossilsOpen discussion area.Gems and GemologyOpen discussion area.GeneralOpen discussion area.How to ContributeOpen discussion area.Identity HelpOpen discussion area.Improving Mindat.orgOpen discussion area.LocalitiesOpen discussion area.Lost and Stolen SpecimensOpen discussion area.MarketplaceOpen discussion area.MeteoritesOpen discussion area.Mindat ProductsOpen discussion area.Mineral ExchangesOpen discussion area.Mineral PhotographyOpen discussion area.Mineral ShowsOpen discussion area.Mineralogical ClassificationOpen discussion area.Mineralogy CourseOpen discussion area.MineralsOpen discussion area.Minerals and MuseumsOpen discussion area.PhotosOpen discussion area.Techniques for CollectorsOpen discussion area.The Rock H. Currier Digital LibraryOpen discussion area.UV MineralsOpen discussion area.Recent Images in Discussions
Techniques for CollectorsCleaning native copper

17th Feb 2006 00:09 UTCKyle Eastman
-Kyle
17th Feb 2006 03:39 UTCPaul Brandes 🌟 Manager
Are you sure it's malachite, or just the green tarnish that forms on copper??
One of the most common (and inexpensive) ways is to go to your local hardware store and buy muriatic acid and soak the pieces in that. However, this method is best used when there are NO crystals present and you don't really care about anything except removing tarnish or if you want to remove any carbonate (calcium, etc...) from the piece. If you are cleaning pieces with crystals, by all means, don't!! You run the risk of damaging the crystal faces and ultimately ruining the asthetic quality of the piece, at least in my opinion. I know you can also use sulfamic acid (coffee pot cleaner) to clean copper, although it is a much slower process it's also a much safer one to the copper; not as likely to etch.
CaptPaul

17th Feb 2006 15:39 UTCKyle Eastman
I am fairly sure it is malachite, but not 100%. I have actually tried muriatic acid, and it cleans fairly well, but some sort of coating always seems to return after I take the specimens out of the acid. I will not etch pieces with crystals until I have a reliable techinque to do so. That coffee pot cleaner idea sounds like a good one.
-Kyle
17th Feb 2006 17:44 UTCDavid Von Bargen Manager
17th Feb 2006 20:41 UTCPaul Brandes 🌟 Manager
What we always did in Michigan after we cleaned our copper was to dip the pieces in something called "Copper Brite", which is a mixture of chromic acid and other chemicals. What this does is put a real thin coating on the specimen to keep it from oxidizing again.
Another trick to clean copper (and silver too) is to dip it in TarnX, which I have found won't etch crystals. If you can't find any of these chemicals, another old trick is to simply rub the crystals with your fingers providing you can do so without cutting yourself ;-)

18th Feb 2006 19:14 UTCKyle Eastman

23rd Feb 2008 18:54 UTCBarry Strieter

24th Feb 2008 04:39 UTCAlfredo Petrov Manager
(Removes apatite from teeth too, in case you want to switch to ceramic teeth.)
28th Mar 2008 11:35 UTCRock Currier Expert
Rock

10th Aug 2008 19:25 UTCJohn Kupar
Best Regards,
John
10th Aug 2008 20:05 UTCŁukasz Kruszewski Expert
Good luck!

10th Aug 2008 22:16 UTCJohn Kupar
Thanks!
John
10th Aug 2008 23:45 UTCPaul Brandes 🌟 Manager
Please read my earlier posts (CaptPaul) to get a start on cleaning your UP coppers.
You must have been there for the Mineral Retreat?? What 3 mines did you visit??

11th Aug 2008 01:21 UTCjeff melanson

11th Aug 2008 02:16 UTCCostas Constantinides
This is what I do and it works well on small pieces but when specimens are large and very dendritic it's just good.
The idea is to get the acid and water off and dry as fast as you can.
1..remove specimen from acid and rinse in water
2..now rinse specimen in a container of warm water thoroughly
3.rinse the specimen in metholated spirits.This enables the remaining water to mix with the metho and when removed to dry does so quickly.
4.lightly coat the specimen with your choice of copper type varnishes
I'm not fond of doing this as most native coppers are best preserved as they are
Costas
7th Dec 2008 02:26 UTCKeith Compton 🌟 Manager
Sorry I didn't see your message earlier.
I don't like to clean any "green" off native copper - whether natural oxidisation or any of the many copper mineral coatings.
However, given your question here are two tips:
(i) I don't think you can get TarnX in Australian but you can use Goddards Silver Dip - you can generally pick this up in the cleaning sections of Coles or Woolies or any hardware store. You could also look at some of the car wheel cleaners - some of these have similar principal chemicals.
(ii) Rather than use a copper varnish you could consider hair spray - much easier to apply and more importantly, to remove.
Cheers

11th Jan 2009 08:34 UTCFranklin Roberts
The original process involved placing the copper piece to be cleaned into a bath of saturated sodium cyanide solution, adding a slug of 30% hydrogen peroxide to the container in one portion and then running like hell. Within a few seconds, the mixture would flash to steam, ejecting a cloud of boiling hot water, cyanide solution, steam, oxygen and a cloud of deadly hydrogen cyanide gas into the air and all over the immediate surroundings. For the life of me, I cannot imagine a more foolhardy way to clean and polish metal. For somebody to take such a stupid risk, there had to be some sort of payoff at the end.
There was. Not only did the the process instantly remove hard, pitted corrosion, it actually imparted a mirror-bright polish to the workpiece. I can only assume that it did so by first reducing the oxide coating and then removing a layer of copper a few atoms thick. I've seen the results of the cyanide bomb technique and it looked as if somebody had invested several hours of elbow grease instead of a few seconds time (and half a lung).
I had read somewhere that thiourea, the main ingredient in Tarn-X, was finding many new industrial uses, mostly replacing cyanide in some metallurgical processes such as leaching and plating. I had access to a lab and the proper chemicals at the time, so I attempted to duplicate the cyanide bomb process by simply substituting thiourea for sodium cyanide. I was pleased to discover that the experiment worked perfectly on the first attempt. While it was still a violent exothermic reaction and very messy, at least there was no cyanide to clean up. Even so, this reaction is an "outdoors only" experiment.
The process imparted a beautiful mirror shine to several non-ferrous metals that I bombed, even to stainless steel.
Frank

11th Jan 2009 11:25 UTCAnton Ivanov
regards
Anton

13th Oct 2010 09:33 UTCandrew
13th Oct 2010 13:58 UTCReiner Mielke Expert
The results also depend on the impurities in the copper, some Michigan copper will not clean at all. The best thing I have found to clean copper (in that it stays cleaner longer afterward) is using citric acid. However even then you can expect it to tarnish brown in a couple years if not sealed with something.
22nd Oct 2010 18:38 UTCPeter Haas
Also note that thiourea is an extremely strong mutagen that should be handled with as much care as possible ! Denaturing proteins with thiourea is easy as pie. Don't let the name fool you - the relation to urea is only a structural one, its properties are quite different.

10th Jan 2012 06:11 UTCAnonymous User
10th Jan 2012 09:42 UTCRock Currier Expert
10th Jan 2012 13:47 UTCPaul Brandes 🌟 Manager
First off; welcome to Mindat!!
Now as far as cleaning your copper, I see two mistakes immediately. First, never leave the copper in muriatic acid overnight! This will “burn” the copper and destroy any crystals you have. I usually only leave any specimens in for 5 minutes at the very longest, and then only if there are no crystals on it (I do not use muriatic or any strong acid on crystalised specimens). Second, you are trying to do your cleaning in mass (25-50 pieces at a time); this is way too many at once. What is more than likely happening is you are putting so many pieces in that you are diluting the acid which changes the chemical makeup of the solution and will result in funky coloured pieces of copper. I will only do one or two at a time and if there is a lot of material to be removed, I will not re-use the acid, but rather start another round with fresh acid. And then there are just some pieces of copper, especially the higher arsenic content coppers from the Keweenaw Peninsula of Michigan, that you will have a hard time cleaning. It can be done, but with acids I don’t recommend using. Many of the folks in the Keweenaw will use a combination of acids and other chemicals to clean copper, and then as a final step, use Copper Brite (the acid dichromate solution Rock mentioned) to preserve the finish. But as Rock also mentioned; most serious collectors of native copper leave their specimens in a “natural” state with the patina intact.
If you just want to clean your copper, I would recommend placing them (2 or 3 at a time) in muriatic for no more than 5 minutes, then rinsing them off with water (a small pressure washer works well for this), drying them (I generally use a heat gun or hair dryer), then dipping them in Copper Brite. If you have crystalised specimens, I would leave them alone as most acids will ruin the sharp crystal edges by rounding them off...
10th Jan 2012 17:11 UTCScott Sadlocha
My problem is that I don't like the shiny "new" copper appearance. My favorite coppers are those that have the brownish patina to them, almost as if they had been kept in someone's pocket for a few decades. I have a decent amount of specimens like that, I am not willing to soak them in anything. Still, quite a few of them would exhibit more crystallization if I could find some way to clean them better. In some cases, the copper disappears into fractured calcite, or into matrix rock, but I can tell that there is more crystallization evident based on a few exposed surfaces.
I have tried mechanical cleaning, using small picks and a water gun. This does work in many cases, but it seems to take forever. On some pieces, it seems like it will take me 20-30 runs with this method to clean things out. The worst cases are those with copper and distinct calcite crystals, with matrix I want to remove. The copper is durable for the water gun, but I have to really be careful near the calcite.
When I get home, I will try to post a photo of what I am talking about. One piece that I have found particularly vexing falls into this category, and the piece almost seems to be wrapped with a granular matrix. I talked to some of the guys from the Caledonia mine at the last Detroit show about this piece, and they suggested continuing with the mechanical method. I think a lot of the pieces I have are just going to have to wait until I have a heck of a lot of time to work them over. I do have one piece at home that I really like, but I am not sure how it was cleaned. It does have a bit of a "new" look to it, but it is not overly shiny. More of a matte finish, and the crystallization is fantastic, though it is just a TN. Unfortunately, I can't find out how it was cleaned, as I found it at an estate sale quite a while back, and the original owner was not available.

10th Jan 2012 19:07 UTCAnonymous User

10th Jan 2012 19:12 UTCMichael Hatskel
It is not supposed to be happening, is it?
Dilute non-oxidizing acids, like HCl, at room temperature shall dissolve the surface coatings (oxide, sulfide) on copper but not the metal itself. Unless there is a sizable amount of a more electro-positive impurity (Ag, for example) that helps copper to corrode. But that is less likely to happen in the well-formed copper crystals.
Does someone have an explanation of the observed copper dissolution? What am I missing here?

10th Jan 2012 21:14 UTCJoseph Polityka Expert
You can try this method I was taught years ago by my high school science teacher. He showed me this cleaning technique on a copper penny which you might want to try first.
Place the tarnished penny in a suitable glass container. Cover the penny with white vinegar. Drop a small quantity of ordinary table salt into the vinegar and watch the penny magicallly turn bright and clean. Safely dispose of the solution and start again on one of your small copper specimens.
Let me know what happens, please.
Good luck,
Joe

11th Jan 2012 03:44 UTCAnonymous User
11th Jan 2012 09:11 UTCRock Currier Expert
12th Jan 2012 03:22 UTCScott Sadlocha
This first one is the small TN I was referring to. It is about 3cm x 2cm, and has a bit of an iridescent sheen to it, and I believe it was chemically cleaned. Still, the crystals are pretty sharp, so however it was cleaned was a decent job. I am not very fond of the finish of the piece, but otherwise I really like it. The branching nature of the piece, along with the crystallization (and what I believe might be twinning) make this one of my favorite coppers.

12th Jan 2012 03:36 UTCAnonymous User

12th Jan 2012 03:39 UTCAnonymous User
12th Jan 2012 03:40 UTCScott Sadlocha
This second one is a bit larger, but is an example of a piece I would like to clean a bit, but not change. It is about 25cm x 13cm, and I was told it came from "York Mine" or "New York Mine". Since I can't match that up exactly with an existing mine and there are several mines and prospects that reference New York and various parts of it (Brooklyn), I can only be sure it comes from Copper Country. This piece is an example of one that I would like to keep relatively intact. I really like the red and green patina to it, but would like to clean the gray material out of all the nooks and crannies. This is one that is taking some mechanical cleaning to get it looking better. Besides the green and red staining, there are some spots of light blue on it, though I am not sure what it is (the color is not right for chrysocolla or something along those lines) and there are some micro pockets of epidote on the bottom. This piece is an example of quite a few I have--where most of the dirt is gone, but there is some tough, hard material stuck in small pockets.
12th Jan 2012 03:46 UTCScott Sadlocha
I just noticed that Bill is posting as well. A lot of what he has is similar to material that I have, where quite a bit more cleaning is needed. On some of the pieces, you can tell there is copper crystallization, but it is hard to see with all the matrix making it look like a lump.
*This last piece is from the Indiana Mine.

12th Jan 2012 04:02 UTCAnonymous User


12th Jan 2012 21:44 UTCPaul Brandes 🌟 Manager
Did you collect the Phoenix specimen yourself? Reason I’m asking is are you sure that’s a Phoenix specimen? To me, it looks a lot like the Laker Pocket coppers that came out in 2008 which would have a brick red patina naturally. Your Indiana crystal is typical of what comes from there; partial/modified crystals with a tenorite coating.
The “York” piece and all of yours Bill are more in line of what comes out of the piles these days. One thing I failed to mention before is for the hard to reach spots, a can of Dow Scrubbing Bubbles does a pretty good job of loosening up the gunk without damaging anything, then water gun or pressure washer clean it the rest of the way. I’ve had pretty good success with this method. Another item that works quite well are the small spring loaded centre punches you can buy at Harbor Freight or Northern Tool. A few repeated pops usually will loosen the rock to a point you can chip it off fairly easily. Just be careful you don’t hit the copper as it will damage it quite quickly. As far as acids, muriatic is probably the safest and best “stronger” acid to use.
Bill, do you have any idea where your coppers are from (what mine)?? It would appear that a couple of those, especially the one in photo 010, are somewhat crystalised so you might want to be a little careful with those; the rest I would dip in muriatic for a couple minutes, remove, rinse, and repeat until you get the desired look. Some of those look like they will clean up nice with a little patience; others, I wouldn’t hold my breath on…..
13th Jan 2012 08:37 UTCRock Currier Expert
I don't think you are going to find any chemical that is going to make all those specimens look really good. A little hydrochloric acid will remove any calcite that is on the specimens, but it will also probably remove any tarnish or oxidation that you may find attractive on the specimens. Then what you will most likely be left with are silicates like quartz, epidote and rock. The only way to effectively remove those would be through the use of Hydrofluoric acid and I am not sure I see any specimen in your group photos what would be worth the danger and expense of using that reagent. Even then about the best you could hope for is to strip the specimens down to the bare copper and have it really bright. Of course with time the color will tone down and oxidize. But of course there is no guarantee that they will oxidize to the color and or tone of your taste. You can experiment with fresh bright copper in a variety of ways like holding it over a flame and you may hit upon a method that will oxidize it just the way you want, but I won't be able to advise you there.

13th Jan 2012 15:21 UTCAnonymous User

13th Jan 2012 15:56 UTCD Mike Reinke
Ain't it great?! And the more you learn, the more enjoyable it gets. The crust of the earth is amazing...deep, in more ways than one. Having your wife share the interest is the best. I love my rock collection, but it can't love me back, can't even get coffee...
Type 'epidote' into mindat, you'll see plenty in the gallery. keep you occupied this winter.
Have you tried sandblasting a copper piece? I had an acid damaged galena that was expendable, it looked better sand blasted (w/o my glasses on). It is too soft, it looks pocky upon closer inspection but it took the crud off. Copper would fare better.
Take care,
Mike
13th Jan 2012 21:25 UTCRock Currier Expert
Can you be more specific about the photos you are talking about? Epidote is a green to black mineral often found associated with copper specimens from Michigan.
14th Jan 2012 01:56 UTCPaul Brandes 🌟 Manager
Just a suggestion...

14th Jan 2012 18:30 UTCAnonymous User
14th Jan 2012 22:32 UTCScott Sadlocha
Bill, you can add a photo in your post once you attach the file. There will be a button that title "Create Link in Message" once the file is attached to the message. What I usually do is type up what I want to add to a post, add the files, and then put my cursor down below my text a couple lines. I then click on the button to add the photo to the post (you won't actually see the photo until the post is made, but there will be a text line), drop down a couple lines, and do it again, repeating for all the photos.
Paul, I did not collect the Phoenix piece myself, so I am not entirely certain. I got it from the estate sale of a collector, and that is the information I was given at the time. I would much rather it be a Laker pocket piece of course, but really couldn't say, and I am not sure how that could be determined. Either way, I like the piece a lot, and even though it is small, it is one of my favorite coppers out of the many I have. There is just something about it that really appeals to me.
15th Jan 2012 00:49 UTCPaul Brandes 🌟 Manager
It's not my decision Bill what can be discussed on here, that's for the managers. It was more of a suggestion that if you wanted to chat about other things besides how to clean copper to start a locality talk page on the Copper Country, and there you could really have a grand discussion about all things Keweenaw. :-D
Earlier you were asking about epidote; here is an example from the Centennial No. 2 Shaft in Houghton Co.

15th Jan 2012 04:18 UTCBart Cannon
Copper and silver are sulfur getters. They draw sulfur out of the air.
I wonder is a quick flash in diulte nitric acid might produce a tolerable and not overly bright finish.
I've used "Linde A" polishing compound applied and rubbed with a stiff artist brush to produce a "natural" looking clean-up on both native copper and native silver.
15th Jan 2012 05:37 UTCScott Sadlocha

15th Jan 2012 07:54 UTCJim Gawura
I've noticed that some copper when being rinsed in water to wash off any remaining acid will oxidize if left in the water for a day or more. It appears that whether or not the oxidation is fast, slow, or non existent depends on the chemical makeup of the copper. I've had specimens that had to be taken out of the rinse and either blown or sun dried immediately or they would oxidize. Others could soak for days and still come out bright! Same in the cabinet, some of the copper is fairly oxidized after only in a few months and other pieces have been in the cabinet for years and are still shiny. Also had both results from the same mine. I use sulfamic acid exclusively for any acid cleaning. Natural and just washed and scrubbed or pressure wash is my preferred cleaning method. Unfortunately it seems that the majority of the specimens I collect have some kind of nasty coating that usually has some calcite next to the copper. Acid works fine. My experience has been that HCL no matter how dilute and how short a duration the soak it etches copper crystals and rounds off all the sharp edges of the crystals. Sulfamic undoubtedly etches the copper, but at a rate so slow I can not discern any damage. Sulfamic acids main use is for cleaning out boilers. Usually comes as a white powder in 50lb. bags.
Good Luck,
Jim

15th Jan 2012 15:05 UTCAnonymous User
15th Jan 2012 19:02 UTCScott Sadlocha
I can't sing the virtues of this cleaning enough. I have more than a handful of specimens that elicited "Oh my gosh!" moments after I gave them a go with the water gun, including one especially funky Siderite with Pyrite from Eagle Mine. It is really great for copper because you don't have to worry much about being too careful, unless there are accessory minerals. Quite a few people and dealers sell them, and you should be able to find them fairly easily. I bought mine for about $80 a few years ago, and it has performed flawlessly since.

16th Jan 2012 02:27 UTCAnonymous User

16th Jan 2012 02:54 UTCD Mike Reinke
Scott's idea is probably better than sand blasting, but ran into a guy who was strapped financially, but had some ingenuity; He took stuff he found, larger pieces anyway, down to the carwash, secured them some way, then used that power wand on 'em! i got a kick out of that. A country boy can survive I guess...
Mike

16th Jan 2012 18:22 UTCAnonymous User
16th Jan 2012 18:42 UTCScott Sadlocha
On this piece, I can see some decent copper crystallization peeking out, as well as a few decent crystals already exposed. The first shot is of the whole piece, what I would consider the front, and the second is a closeup. Subsequent posts will show other angles and more of the matrix. As you can see on these shots, there is some calcite attached. While the periphery calcite is damaged, there are some very nice smaller crystals in the center, and I would rather not damage these. I am not sure if it can be seen, but there are a few areas with a coating of micro-crystalline quartz as well. This entire piece is about 10cm x 10cm.
16th Jan 2012 18:49 UTCScott Sadlocha
This second set of photos shows how the matrix material wraps around the copper. It is almost like the copper was encased in it (and it most likely was if my assumption is correct). At points where they meet, there is actually a small gap which I think I may be able to exploit to separate them. There is some layering extending outward, including a thin layer of pistachio green epidote where the matrix meets the copper. This matrix is very porous and somewhat crumbly, and peppered with very tiny crystals of various accessory minerals, especially in the larger bubble remnants. Based on the porosity of it, I thought it would break off a bit easier than it has. Still, with several goes from the water gun, I have been able to whittle away at it.
16th Jan 2012 18:52 UTCScott Sadlocha
16th Jan 2012 18:58 UTCScott Sadlocha
16th Jan 2012 19:03 UTCScott Sadlocha
16th Jan 2012 19:06 UTCScott Sadlocha
Again, I don't have in front of me the location. These are both smaller pieces, like the Phoenix piece.

16th Jan 2012 23:21 UTCBart Cannon
When we came across Keeweenaw coppers that were enclosed in basalt, we would heat them up in the oven at highest heat, and them drop them into a bucket of cold water. Many treatments were necessary, but with luck, most of the basalt would fall away. No acid, No neutralization needed.
It was thus possible to convert a worthless specimen into a five dollar specimen via ten dollars of effort.
Bart
16th Jan 2012 23:56 UTCDan Fountain
I like your idea a lot. Sometimes we'd find that just using a hammer to shatter the basalt was the only effective means, but the thermal method seems a lot more elegant - plus you get to heat things up way beyond safe limits & throw them in water! R-R-R! More power!
17th Jan 2012 02:52 UTCPaul Brandes 🌟 Manager
-------------------------------------------------------
> I don't think that this cleaning method has been
> mentioned.
>
Actually, it has been mentioned in many books and research papers over the years. This was the method used over 5,000 years ago by the ancient miners of the Copper Country who wanted to separate copper from basalt. It is said that they would build a large bondfire along a stream or lake and toss copper into it to heat it up. Once hot, they would throw ice cold water on them and supposedly, the contraction of the copper would shatter the rock, sometimes with explosives results. I am told that it does work, although it's not the most effective way to remove basalt from copper.

17th Jan 2012 06:54 UTCBart Cannon
I'm on the edge of my chair. What IS the best method to remove basalt from Michigan coppers ?
One method I've thought about is known as the "electromagnetic pulse pulverizer". I worked with a guy who was developing it to liberate diamonds from kimberlite. He was able to liberate orthojoaquinite crystals in fine grained syenite for me from a rare earth deposit in Alaska.
He went on to different dreams, and told me how to build the device. It required high voltage capacitors the size of oil drums and a the switch was driven by a bullet. More hazardous than the bucket steam bomb I once employed on coppers.
I have done much research on ancient indian mining in the Keeweenaw. I even worked with Dr. Rapp on the trace element chemistry via WDS on Michigan coppers with the hope that we could establish trade routes and copper sources for copper artifacts in North America. I had crazier hopes. I wanted to verify Pre-Columbian trade contact between Europe and the Keeweenaw people.
We determined that trace chemistry was hopeless via WDS, and even hopeless via mass spectroscopy because the chemistry of Michigan coppers changes by the foot in the mine. My final conclusion was to use silicate inclusions as fingerprints, but no European museum would allow messing with their artifacts.
Much the same with another of my projects. Determining the provenance of the spear point in Kennewick Man's ilium. Curators mostly just want to keep stuff pristine in their museum drawers. When I was examining K-Man's ilium in the Burke Museum with my Optivisor, hands behind my back, I got tapped on the shoulder and told "you are too close".

17th Jan 2012 12:55 UTCAnonymous User
18th Jan 2012 01:46 UTCPaul Brandes 🌟 Manager
-------------------------------------------------------
> Paul,
>
> I'm on the edge of my chair. What IS the best
> method to remove basalt from Michigan coppers ?
>
Unfortunitely, one of the best ways to remove basalt is also one of the most dangerous; that's to use HF, which I do not recommend anyone using!! As I mentioned before, one of the spring loaded centre punches works well, as does just carefully chipping away at the basalt. I know that can be time consuming, but at least you'll live to see tomorrow.
Bill, I believe what Bart is talking about are the different coloured basalt matrix one can find attached to the coppers of the Keweenaw. They are actually quite easy to distinguish; green is epidotised basalt. Black is the common colour for basalt. A chocolate brown/reddish coloured basalt is where the majority of the "good" minerals were found during the mining days. The Pewabic Lode in Quincy and other mines nearby was this reddish/brown colour and it was a sign to the miners that they were in potentially good ground.

18th Jan 2012 03:40 UTCAnonymous User

18th Jan 2012 05:23 UTCD Mike Reinke
Bart has not answered, and I've never tried it, but I'll keep it for future reference, certainly. My, and I guess most ,ovens hit 550 degrees. That wont melt copper or basalt, so crank it...How long, don't know, but I bake bread for 50 minutes, and I would suspect a rock if small, would be pretty hot in 50 minutes, or less, of course, depends on the rock... And certainly, drop in water ASAP, I'd guess, or you lose heat. Winter would be a good time to do that, heat your house. Summer's hot enuf w/o that...
Mike

18th Jan 2012 06:04 UTCJim Gawura
Your Phoenix specimen looks like most of the pitting and rough surface may be do more to 150+ years of oxidation on the pile then the acid. To long in the hcl will give the specimen a melted look. If the piece wasn't totally enclosed by calcite it's probably oxidation. A lot of Keweenaw copper has a thin layer of calcite around it and etching away the calcite will leave that small gap that can be exploited in removing the matrix. Unfortunately with some specimens it's almost as if the matrix is part of the top layer of copper. Specimens from the Seneca #1 have been the most frustrating to clean for me. You might reduce or eliminate the iridescence on the acid cleaned copper by quickly drying it once out of the final rinse. My results have been mixed. Some copper appears more susceptible, but a quick blow off and follow up with a heat gun, from prior incidents don't let your wife catch you with her hair dryer, or placed in direct sunlight on a warn day seen to help. Tarn-X is made by Jelmar in Skokie Illinois. The MSDS shows it it contains Sulfamic Acid and Thiourea, but the mixture is listed as proprietary. If your interested in obtaining Sulfamic Acid or some other tips PM me. Spent 30 years collecting in the Keweenaw.
Cheers,
Jim

18th Jan 2012 07:15 UTCBart Cannon
My copper baking was done in 1963 and 1964. I was 13 years old. The oven was also about the same age. I don't remember paying any attention to the operating conditions, but I suspect 300 degrees would be enough to get the rock hot enough for thermal shock and plenty of differential expansion of dissimilar materials.
At 50 cents per 50 cent word, I think you now owe me $2.00.
As for HF, I would be dis-inclined mostly because of the expense of the HF versus the value of the copper specimen.
In previous decades I frequently used HF and only suffered one wound. Somehow a drop found its way to my cuticle and a day later that area felt like a bruise. One tip about HF is that you should never use rubber gloves. They give you a false sense of protection, and they always have a pinhole leak. Tongs are the only way, but you still run the danger of a splash
If you use HF you should always have a tube of calcium gluconate antidote gel very handy.
My use of HF declined precipitously after I bought a CRC tome on lab safety which had dozens of color photos of HF wounds. The ones showing melted eyeballs are quite life changing.
Bart
18th Jan 2012 11:59 UTCPaul Brandes 🌟 Manager
-------------------------------------------------------
> Specimens from the Seneca #1 have been
> the most frustrating to clean for me.
That's not surprising, considering the copper in that area of the Keweenaw around Seneca/Mohawk has a higher concentration of arsenic than most places. No matter what one does in attempting to clean a specimen from there, it never really comes out great.

18th Jan 2012 23:56 UTCBart Cannon
Seems that it's Bill Boehm who owes me $2.00 for my 50 cent words, not Mike.
Hard to keep track, isn't it. I don't believe that it was me who mentioned "green, black, brown rocks" enclosing coppers.
I do have another suggestion about cleaning coppers that would require $25 dollars of effort to produce a $5 copper.
My EWL air abrasive station has, in addition to its bead blast guns, a very powerful micro jack hammer. It will remove chunks of rock when set to high pressure, and can work more delicately at lower pressure.
Still not an economic solution for coppers, but there is something therapuetic about punishing matrix.
Bart

19th Jan 2012 02:18 UTCAnonymous User

19th Jan 2012 02:49 UTCAnonymous User
Paul: I scrolled through the nine talk pages and there wern't any topics about the Keeweenaw that I could add to. I could not find a way to start a new post. Can someone tell me how to start one? Thank you.

19th Jan 2012 03:47 UTCD Mike Reinke
I'm new enough at this too, but I'd venture to say you can put any rock in an oven. Lava runs in the vicinity of 2000 degrees F, give or take, right? So don't worry, you've got lots of leeway there. Some cherts, I hear, have moisture in them and don't make good campfire rocks because they can blow up, but i have never known of that happening. If others could weigh in on that i'd like to hear...You are talking at most fist sized pieces in an oven, aren't you, I'd guess. Baking a turkey sized hunk of pure arsenic may not be wise, but the little in copper, I can't see any danger, IMHO.
I also don't think there are too many minerals that would ever need, or could withstand, this kind of abuse. horrible cruelty!
There was a great thread on safety just recently. Go to the home page, upper right,and at 'search for' scroll to messageboard and type 'arsenic and cinnabar' and that thread will come up. It'll put your mind at ease. Mineralogy is not extreme sports.not usually.
Muriatic acid is dicier stuff. I only use it outdoors.
Bart, great comment on 'punishing matrix', so true!
Mike
.

19th Jan 2012 04:33 UTCAnonymous User

19th Jan 2012 05:35 UTCD Mike Reinke
Localities might work better if you give it less. i found that using one key word would get me close, then i could scroll though what came up, i.e. type in just a county, then when all the states came up, scroll to michigan and you might find what you want. Managers could help more, but this has got me through .
mike

19th Jan 2012 15:28 UTCBart Cannon
I would avoid oven treatments of mercury ores, thallium ores and realgar bearing rocks. I don't think that arsenopyrite will release arsenic oxide fumes at 300 degrees F.
There are extensive discussions about the dangers of minerals someplace on the website.
Good news. I've decided not to bill you for my 50 cent words. Just reading them is enough penance.
Bart
19th Jan 2012 16:45 UTCDavid Von Bargen Manager
http://manual.mindat.org/index.php/Talk_Pages

19th Jan 2012 16:46 UTCAnonymous User

22nd Jan 2012 15:45 UTCAnonymous User
22nd Jan 2012 17:18 UTCDavid Von Bargen Manager
You cannot start a new topic for the talk pages from the fora, they need to be started from mineral or locality pages.
22nd Jan 2012 20:45 UTCPaul Brandes 🌟 Manager
I'm sorry you are having issues with the site, but please don't be discouraged. Remember, everybody at one time was in the same situation you are in now and by trial and error, you will learn how to use the site. I've been coming here for many years and I still learn new things about the site.
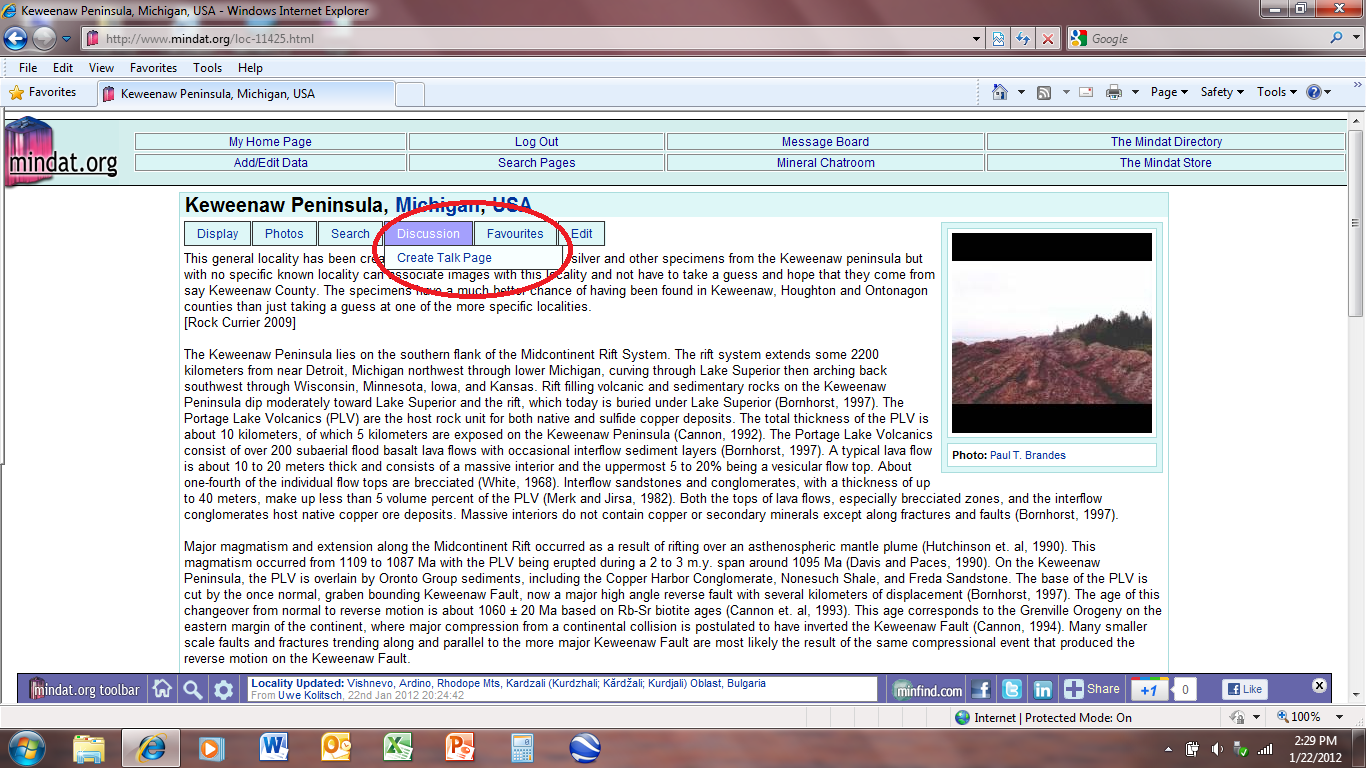
Now to answer your question about the talk pages: I attached a photo below to show what you should see to create a talk page for the Keweenaw Peninsula. You should be able to see the toolbar under the locality name. Highlight the "Discussion" tab to make the "Create Talk Page" drop down appear. Just click on that and you should be able to create a talk page for the Keweenaw or any other locality or mineral.
22nd Jan 2012 22:24 UTCScott Sadlocha
Try not to get too discouraged. Once you navigate the site a bit, you will get the hang of it a bit more. There is a lot of functionality here and it takes some getting used to. Also, one other thing I should mention--most of the time, folks here are fairly good at responding. However, this time of the year, you might not get a response as quickly as you normally would. Right now a lot of folks are getting prepared for, or are already in, Tucson, Arizona, for the largest mineral show in the world, which happens yearly at the end of January and beginning of February.
7th Oct 2012 19:51 UTCScott Sadlocha
1. With a vinegar cleaning method, is the vinegar used full strength or diluted? I have heard it takes longer to soak and clean. How long should it take?
2. Will a sulfamic soak remove calcite or just do more of a cleaning?
3. Will any of these acid bath types of cleaning (HCl, sulfamic, vinegar) damage pumpellyite?
The reason for my questions are the specimens I brought home. I have quite a few pieces with infilled veins where there is much prehnite present, and I suspect a decent amount of copper. I need to remove the calcite infilling, but I don't want to damage the copper. If it comes out shiny, that is fine, I just don't want that "burnt" look that I have experienced with HCl. I have a few pieces of this type of prehnite/copper that others have cleaned, and it is apparent the calcite was dissolved, but they look very nice. I am confident that I have at least a few pieces like this and want to get them opened up.
I also have one miniature sized piece with a very small pocket on one end of it. Looking at the edge of the pocket, it looks very much like radiating pumpellyite. However, beyond this the entire pocket is infilled with calcite. I want to remove it and not damage anything else.
I also have some pieces with native copper, and I need to spend some time trying to figure out how to clear it out, but I am focusing on the two types of pieces I mentioned above first.
Thanks!
9th Oct 2012 19:04 UTCScott Sadlocha
10th Oct 2012 12:43 UTCDan Fountain
I'll try to answer from my limited experience.
1. Generic white vinegar in the US is usually 5-8% acetic acid (sez Wikipedia). That's pretty dilute already; I don't see any reason to dilute it more.
2. Sulfamic acid will definitely remove calcite. I've used concentrated and dilute - usually using concentrated (saturated solution) when I'm not expecting to find copper under the calcite and diluting a saturated solution about 4:1 if I'm removing calcite from copper. Whatever you use, check it often - starting at every few minutes, to every few hours, to several times a day until you get a feel for how fast it's working on your particular material. I think that rinsing has been stressed in this thread before...
3. Dunno.
I've left some copper crystals that I have found free of matrix in their native state with patina - I just remove dirt and surface grime with detergent and a soft brush. The "scrubbing bubbles" produced by a metal catalyst such as copper in hydrogen peroxide do a good job of cleaning without changing the surface. This also works for cleaning red hematite mud from those delicate needles of pyrolusite, but it may darken some of the bright silvery crystals slightly.
Hope this helps. Whatever you try, don't do your best specimen first.
-Dan
10th Oct 2012 17:46 UTCScott Sadlocha
Since the pumpellyite is a silicate, I am not expecting any issues with a soak, but I want to make absolutely sure before doing so. I am really interested to see how this would turn out, and I only have one specimen like this.
I know what you mean about cleaning with a brush. This is my first step, and I usually evaluate after this to see if this might be all that is needed. With those pyrolusite specimens we collected, I think that is all that will do. Some of those are absolutely stunning after a good cleaning. I think the rhodo we collected (altered or coated) is going to get a blast from a cleaning gun to clean out the crevices. I will let you know how it goes.
10th Oct 2012 18:28 UTCPaul Brandes 🌟 Manager
I saw your question on Oct. 7 but haven't had the opportunity to respond; that little thing called work always seems to get in the way of fun activities!!!! :-(
Anyway to your questions:
1) Pretty much what Dan stated. Normal white vinegar you can buy at any grocery store will work, but it is slow and as long as you can handle the acetic acid odour you'll be fine.
2) Sulfamic will dissolve any carbonate, including calcite; however, I have found that cold sulfamic acid will not dissolve the calcite as aggressively as warm or even room temperature acid, and not nearly as fast as HCL. As Dan stated, the key to using sulfamic or any acid is to babysit the pieces and check them every few minutes (or seconds in some cases) to make sure you don’t remove more than intended. A quick dip and rinse will clean calcite, anything longer and it’s bye-bye carbonate!
3) Pumpellyite is not affected by HCL, so you'll be safe with either HCL or sulfamic. Sulfuric will dissolve pumpellyite slowly, and HF will dissolve it almost instantly. As far as prehnite; be careful placing a specimen in HCL. It will attack the specimen slowly which will whiten any crystals and eventually turn them into powder if you leave them in too long, unless your aim is to remove the prehnite. A quick dip in HCL doesn’t seem to bother them so long as it’s rinsed properly afterwards.
Have fun and post a few pics of the results for us…..

18th Dec 2012 15:27 UTCrufus
have a look at the samples In have - I want to keep them this bright as this is how they came out of the ground !!
19th Dec 2012 08:52 UTCRock Currier Expert
25th Dec 2012 17:40 UTCReiner Mielke Expert

21st Sep 2013 23:20 UTCkishor
i have pure copper sample and i have to do EBSD. but after polishing sample gate corroded and tarnished. so can you tell me method to preserve copper sample after final polishing with collidel silica without affecting crystal structure and plane. so i can do the texture study. thanks
22nd Sep 2013 01:11 UTCReiner Mielke Expert
You could try a light coating of silicon oil or silicon lubricant? Not sure how that will affect EDSD though. Another possibility is to storing the sample in an inert gas such as argon or helium.
20th Jul 2014 20:05 UTCJake Harper Expert
Recently picked up a small collection with some Michigan goodies including these three coppers.
Each one is well xled but they have various coatings that are not the most attractive. I realize strong acids are not an option, however I am wondering if something mild like a diluted citric acid might produce results without a burn? Any help will be greatly appreciated.
Jake
20th Jul 2014 20:10 UTCRob Woodside 🌟 Manager

20th Jul 2014 21:11 UTCAlex Homenuke 🌟 Expert
The first one also looks pretty good as is.
The third one could be improved (IMHO) with careful iterative cleaning. You might try a series of quick dips in phosphoric acid, followed by copious rinsing with water. Another way is using an artists soft brush with the acid to take of enough of the oxidation to show the coppery color, but let it retain some of the patina. There are diverse opinions about cleaning copper.
20th Jul 2014 21:23 UTCPaul Brandes 🌟 Manager
I'm not sure about the first photo, but 2 and 3 appear to be Laker Coppers which, with any type of non-gentle cleaning methods, will fall apart.

20th Jul 2014 21:52 UTCAlfredo Petrov Manager
It also attacks apatite. Guess which mineral teeth are made of? Something to think about while watching the coke do a good job on your coppers :-D
To slow down the tarnishing of copper after cleaning, some collectors and commercial mineral cleaners dip it in a solution of an oxidizing salt, like potassium dichromate, for example, to create a thin protective skin of cuprite over the surface of the copper. The red cuprite film deepens the apparent color of the copper too. The results can be quite lovely but, although Nature herself sometimes coats copper with cuprite, doing it on purpose produces a no-longer-completely-natural mineral specimen. And chromium salts are rather toxic too. I do not recommend this procedure, but be aware that it may have already been done to some native coppers on the market.

21st Jul 2014 02:31 UTCJim Gawura
Cheers,
Jim
21st Jul 2014 05:02 UTCJake Harper Expert
I will certainly go with the consensus and leave these coppers just as they are.
And yet once again the charges of mindat.org save a few more specimens from the impending doom of the uneducated.
Jake

10th Jun 2016 23:47 UTCLarry Hulstrom
11th Jun 2016 02:00 UTCPaul Brandes 🌟 Manager
Your specimen does resemble those that come from the Keweenaw Peninsula of Michigan. One of the first things I would do is check for any copper crystalisation as that will determine what cleaning solutions to use. From your photos, I'm not seeing any crystals so what you have appears to be a normal lump of native copper with attached matrix. With that mixture of matrix (calcite/quartz/basalt), there are few options you can use to clean it. If you read the previous posts by myself and others, that should point you in the right direction.

11th Jun 2016 04:52 UTCJim Gawura
Probably the best way to try cleaning it would be to scrub it and hose it off good. Then submerge it in vinegar, or part of it and see if it fizzes. The vinegar will attack any calcite. It also won't screw up any possible crystals lurking underneath. A real possibility is that you will only brighten the exposed copper. A lot of the ore has basalt directly on it, also quartz, and prehnite are fairly common associations. The acid will not affect any of those. At times a thin layer of calcite will be directly in contact with the copper and if this is etched out you can sometimes break off any other outer coating. Let it soak for at least a week. Check it carefully every now and then and observe it closely for any differences. Vinegar works extremely slow.
Good luck,
Jim

18th Oct 2018 18:18 UTCAllen Wiitala

18th Oct 2018 18:21 UTCAllen Wiitala

18th Oct 2018 18:27 UTCAllen Wiitala
18th Oct 2018 21:51 UTCReiner Mielke Expert

18th Oct 2018 21:59 UTCLarry Hulstrom

20th Jan 2019 14:34 UTCAlex Omanski

14th Jun 2019 16:56 UTCTaylor Vergin
I'd like to preserve the oxide coating while removing the clay and copper hydroxides/carbonates, does anyone have any advice? I've previously used most of the methods listed in earlier posts, but would like to steer clear of acids. The uneven layer of clay allows the acid to remove the oxide on exposed parts of the crystals before it cleans off all the clay from other portions. It seems like there should be chemicals that would dissolve the carbonates/hydroxides and break up the clay without touching the original stable oxide coating. Potentially an oil or oxidizer of some sort?
Thanks,
Taylor
14th Jun 2019 17:30 UTCPaul Brandes 🌟 Manager

15th Jun 2019 03:56 UTCDave Owen

16th Jun 2019 21:58 UTCTaylor Vergin
Here's a photo of what I am dealing with. I'd like to save as much of the original patina as possible, but the clay seems pretty resistant to any sort of physical cleaning. As Dave has dealt with, even soaking the specimens in detergent doesn't appear to work.
16th Jun 2019 23:47 UTCPaul Brandes 🌟 Manager
17th Jun 2019 03:11 UTCSteve Hardinger 🌟 Expert

17th Jun 2019 06:34 UTCGareth Evans
This technique is used essentially in reverse to clean copper. The following formula attacks the black copper oxides but does not attack the copper or the commonly attached cuprite. The solution consists (by weight) of 1 part sodium hydroxide, 3 parts Rochelle salt and 20 parts water. Soak time may be a few minutes to one hour. You will note that the solution turns blue. You will need to clean this off with water – tap or distilled.
I have used it on all my copper specimens without problems. It is a very old method called The British Museum method or the Smithsonian method and it dates back to the early 1900’s. I am surprised that none of the Museum Curators on Mindat mentioned it to you.
Note though that the copper must be free of grit, sand, clay or other material that may hinder the effectiveness of the solution.
I hope this helps!


18th Jun 2019 17:06 UTCSteven Kuitems Expert

20th Jun 2019 22:30 UTCTaylor Vergin
I rotated between brushing on vinegar with a stiff brush and using a high-pressure water gun. It's a lot of effort, but seems to be worth it.
20th Jun 2019 22:50 UTCPaul Brandes 🌟 Manager
20th Jun 2019 23:24 UTCKeith Compton 🌟 Manager
You have done very well .... nice job




Mindat.org is an outreach project of the Hudson Institute of Mineralogy, a 501(c)(3) not-for-profit organization.
Copyright © mindat.org and the Hudson Institute of Mineralogy 1993-2024, except where stated. Most political location boundaries are © OpenStreetMap contributors. Mindat.org relies on the contributions of thousands of members and supporters. Founded in 2000 by Jolyon Ralph.
Privacy Policy - Terms & Conditions - Contact Us / DMCA issues - Report a bug/vulnerability Current server date and time: May 9, 2024 17:25:06
Copyright © mindat.org and the Hudson Institute of Mineralogy 1993-2024, except where stated. Most political location boundaries are © OpenStreetMap contributors. Mindat.org relies on the contributions of thousands of members and supporters. Founded in 2000 by Jolyon Ralph.
Privacy Policy - Terms & Conditions - Contact Us / DMCA issues - Report a bug/vulnerability Current server date and time: May 9, 2024 17:25:06