Boothite
A valid IMA mineral species - grandfathered
This page is currently not sponsored. Click here to sponsor this page.
About Boothite
Formula:
CuSO4 · 7H2O
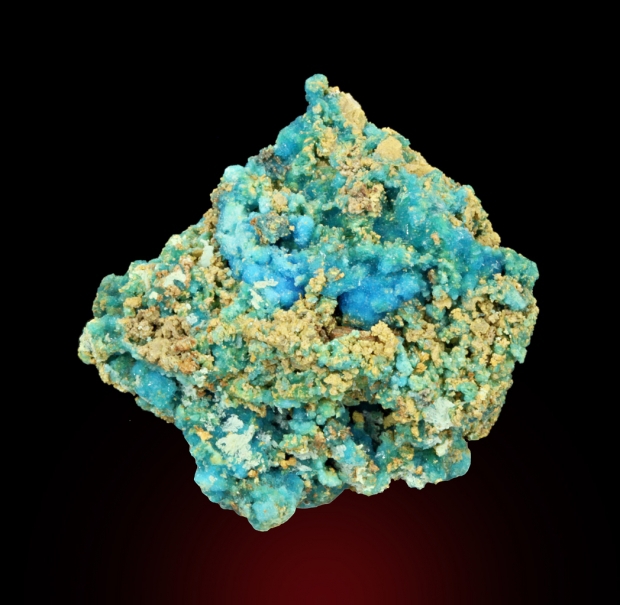
Colour:
Light blue; light blue in transmitted light
Lustre:
Vitreous, Silky, Pearly
Hardness:
2 - 2½
Specific Gravity:
1.94
Crystal System:
Monoclinic
Member of:
Name:
After Edward Booth (July 1857 – 24 August 1917, California, USA) chemist at the University of California. He directed Waldemar Schaller, who described the mineral, to the sulfate occurrence where boothite was discovered.
Melanterite Group.
This mineral, fully hydrated copper sulphate, is very rare. The partially hydrated copper sulphate mineral, chalcanthite, is much more common.
This mineral, fully hydrated copper sulphate, is very rare. The partially hydrated copper sulphate mineral, chalcanthite, is much more common.
Unique Identifiers
Mindat ID:
720
Long-form identifier:
mindat:1:1:720:3
GUID
(UUID V4):
(UUID V4):
da6d9d5b-9014-42a5-b7de-c49e972fa73a
IMA Classification of Boothite
Approved, 'Grandfathered' (first described prior to 1959)
IMA Formula:
Cu(SO4) · 7H2O
First published:
1903
Classification of Boothite
7.CB.35
7 : SULFATES (selenates, tellurates, chromates, molybdates, wolframates)
C : Sulfates (selenates, etc.) without additional anions, with H2O
B : With only medium-sized cations
7 : SULFATES (selenates, tellurates, chromates, molybdates, wolframates)
C : Sulfates (selenates, etc.) without additional anions, with H2O
B : With only medium-sized cations
29.6.10.2
29 : HYDRATED ACID AND NORMAL SULFATES
6 : AXO4·xH2O
29 : HYDRATED ACID AND NORMAL SULFATES
6 : AXO4·xH2O
25.2.4
25 : Sulphates
2 : Sulphates of Cu and Ag
25 : Sulphates
2 : Sulphates of Cu and Ag
Mineral Symbols
As of 2021 there are now IMA–CNMNC approved mineral symbols (abbreviations) for each mineral species, useful for tables and diagrams.
| Symbol | Source | Reference |
|---|---|---|
| Boo | IMA–CNMNC | Warr, L.N. (2021). IMA–CNMNC approved mineral symbols. Mineralogical Magazine, 85(3), 291-320. doi:10.1180/mgm.2021.43 |
Physical Properties of Boothite
Vitreous, Silky, Pearly
Transparency:
Transparent, Translucent
Comment:
Silky or pearly luster on fibrous material.
Colour:
Light blue; light blue in transmitted light
Hardness:
2 - 2½ on Mohs scale
Tenacity:
Brittle
Cleavage:
Imperfect/Fair
On {001} imperfect.
On {001} imperfect.
Fracture:
Irregular/Uneven
Density:
1.94 g/cm3 (Measured)
Comment:
Value approximate.
Optical Data of Boothite
Type:
Biaxial (+/-)
RI values:
nα = 1.470 nβ = 1.480 nγ = 1.490
Max Birefringence:
δ = 0.020

Image shows birefringence interference colour range (at 30µm thickness)
and does not take into account mineral colouration.
and does not take into account mineral colouration.
Surface Relief:
Moderate
Dispersion:
relatively weak
Optical Extinction:
Y = b; X ≃ c.
Comments:
2V(meas.) = Large
Chemistry of Boothite
Mindat Formula:
CuSO4 · 7H2O
Elements listed:
Crystallography of Boothite
Crystal System:
Monoclinic
Class (H-M):
2/m - Prismatic
Space Group:
P21/b
Setting:
P21/c
Cell Parameters:
a = 14.190(10) Å, b = 6.537(2) Å, c = 10.825(6) Å
α = 106.02(5)°
α = 106.02(5)°
Ratio:
a:b:c = 2.171 : 1 : 1.656
Unit Cell V:
0.00 ų (Calculated from Unit Cell)
Morphology:
Crystals rare. Commonly massive with crystalline or fibrous structure.
Comment:
Cell parameters and space group from Leverett et al. (2004).
X-Ray Powder Diffraction
Powder Diffraction Data:
| d-spacing | Intensity |
|---|---|
| 5.358 Å | (22) |
| 5.378 Å | (13) |
| 4.890 Å | (100) |
| 4.826 Å | (43) |
| 2.798 Å | (33) |
| 3.269 Å | (20) |
| 2.730 Å | (11) |
Comments:
Data from Leverett et al. (2004).
Geological Environment
Paragenetic Mode(s):
| Paragenetic Mode | Earliest Age (Ga) |
|---|---|
| Stage 7: Great Oxidation Event | <2.4 |
| 45a : [Sulfates, arsenates, selenates, antimonates] | |
| 45b : [Other oxidized fumarolic minerals] | |
| 47a : [Near-surface hydration of prior minerals] | |
| 47b : [Sulfates and sulfites] | |
| Stage 10b: Anthropogenic minerals | <10 Ka |
| 55 : Anthropogenic mine minerals |
Type Occurrence of Boothite
General Appearance of Type Material:
Fibrous or crystalline massive.
Place of Conservation of Type Material:
n.d.
Geological Setting of Type Material:
Zone of oxidation.
Associated Minerals at Type Locality:
Reference:
Schaller, W.T. (1903) Minerals from Leona Heights, Alameda Co., California. University of California Publications. Bulletin of the Department of Geology: 3: 191-217.
Synonyms of Boothite
Other Language Names for Boothite
Varieties of Boothite
| Iron-bearing Boothite | An iron-rich boothite. Originally reported from Salvadora Mine, Calama, El Loa Province, Antofagasta Region, Chile. |
| Magnesium-bearing Boothite | A Mg-bearing variety of boothite. |
Relationship of Boothite to other Species
Member of:
Other Members of this group:
| Alpersite | (Mg,Cu)(SO4) · 7H2O | Mon. 2/m : P21/b |
| Bieberite | CoSO4 · 7H2O | Mon. 2/m : P2/m |
| Mallardite | MnSO4 · 7H2O | Mon. 2/m : P2/m |
| Melanterite | Fe2+(H2O)6SO4 · H2O | Mon. 2/m : P21/b |
| Zincmelanterite | (Zn,Cu,Fe)SO4 · 7H2O | Mon. 2/m : P21/b |
Common Associates
Associated Minerals Based on Photo Data:
| 2 photos of Boothite associated with Arseniosiderite | Ca2Fe3+3(AsO4)3O2 · 3H2O |
| 2 photos of Boothite associated with Cuprocopiapite | Cu2+Fe3+4(SO4)6(OH)2 · 20H2O |
| 1 photo of Boothite associated with Pyrite | FeS2 |
Related Minerals - Strunz-mindat Grouping
| 7.CB. | Sarvodaite | Al2(SO4)3 · 5H2O |
| 7.CB.02 | Voudourisite | CdSO4 · H2O |
| 7.CB.05 | Dwornikite | Ni(SO4) · H2O |
| 7.CB.05 | Gunningite | ZnSO4 · H2O |
| 7.CB.05 | Kieserite | MgSO4 · H2O |
| 7.CB.05 | Poitevinite | (Cu,Fe)SO4 · H2O |
| 7.CB.05 | Szmikite | MnSO4 · H2O |
| 7.CB.05 | Szomolnokite | FeSO4 · H2O |
| 7.CB.05 | Cobaltkieserite | CoSO4 · H2O |
| 7.CB.07 | Sanderite | MgSO4 · 2H2O |
| 7.CB.10 | Bonattite | CuSO4 · 3H2O |
| 7.CB.12 | Belogubite | CuZn(SO4)2 · 10H2O |
| 7.CB.15 | Aplowite | (Co,Mn,Ni)SO4 · 4H2O |
| 7.CB.15 | Boyleite | (Zn,Mg)SO4 · 4H2O |
| 7.CB.15 | Ilesite | (Mn,Zn,Fe)SO4 · 4H2O |
| 7.CB.15 | Rozenite | FeSO4 · 4H2O |
| 7.CB.15 | Starkeyite | MgSO4 · 4H2O |
| 7.CB.15 | Drobecite | CdSO4 · 4H2O |
| 7.CB.15 | Cranswickite | MgSO4 · 4H2O |
| 7.CB.20 | Chalcanthite | CuSO4 · 5H2O |
| 7.CB.20 | Jôkokuite | MnSO4 · 5H2O |
| 7.CB.20 | Pentahydrite | MgSO4 · 5H2O |
| 7.CB.20 | Siderotil | FeSO4 · 5H2O |
| 7.CB.25 | Bianchite | Zn(SO4) · 6H2O |
| 7.CB.25 | Chvaleticeite | Mn(SO4) · 6H2O |
| 7.CB.25 | Ferrohexahydrite | FeSO4 · 6H2O |
| 7.CB.25 | Hexahydrite | MgSO4 · 6H2O |
| 7.CB.25 | Moorhouseite | Co(SO4) · 6H2O |
| 7.CB.25 | Nickelhexahydrite | Ni(SO4) · 6H2O |
| 7.CB.30 | Retgersite | NiSO4 · 6H2O |
| 7.CB.35 | Bieberite | CoSO4 · 7H2O |
| 7.CB.35 | Mallardite | MnSO4 · 7H2O |
| 7.CB.35 | Melanterite | Fe2+(H2O)6SO4 · H2O |
| 7.CB.35 | Zincmelanterite | (Zn,Cu,Fe)SO4 · 7H2O |
| 7.CB.35 | Alpersite | (Mg,Cu)(SO4) · 7H2O |
| 7.CB.40 | Epsomite | MgSO4 · 7H2O |
| 7.CB.40 | Goslarite | ZnSO4 · 7H2O |
| 7.CB.40 | Morenosite | NiSO4 · 7H2O |
| 7.CB.45 | Alunogen | Al2(SO4)3 · 17H2O |
| 7.CB.45 | Meta-alunogen | Al2(SO4)3 · 12H2O |
| 7.CB.50 | Aluminocoquimbite | Al2Fe2(SO4)6(H2O)12 · 6H2O |
| 7.CB.50 | Lazaridisite | 3CdSO4 · 8H2O |
| 7.CB.52 | Pararaisaite | CuMg[Te6+O4(OH)2] · 6H2O |
| 7.CB.55 | Coquimbite | AlFe3(SO4)6(H2O)12 · 6H2O |
| 7.CB.55 | Paracoquimbite | Fe4(SO4)6(H2O)12 · 6H2O |
| 7.CB.55 | Rhomboclase | (H5O2)Fe3+(SO4)2 · 2H2O |
| 7.CB.55 | Raisaite | CuMg[Te6+O4(OH)2] · 6H2O |
| 7.CB.57 | Caichengyunite | Fe2+3Al2(SO4)6 · 30H2O |
| 7.CB.60 | Kornelite | Fe2(SO4)3 · 7H2O |
| 7.CB.65 | Quenstedtite | Fe2(SO4)3 · 11H2O |
| 7.CB.70 | Lausenite | Fe2(SO4)3 · 5H2O |
| 7.CB.75 | Lishizhenite | ZnFe2(SO4)4 · 14H2O |
| 7.CB.75 | Römerite | Fe2+Fe3+2(SO4)4 · 14H2O |
| 7.CB.80 | Ransomite | CuFe2(SO4)4 · 6H2O |
| 7.CB.85 | Apjohnite | Mn2+Al2(SO4)4 · 22H2O |
| 7.CB.85 | Bílinite | Fe2+Fe3+2(SO4)4 · 22H2O |
| 7.CB.85 | Dietrichite | (Zn,Fe2+,Mn2+)Al2(SO4)4 · 22H2O |
| 7.CB.85 | Halotrichite | FeAl2(SO4)4 · 22H2O |
| 7.CB.85 | Pickeringite | MgAl2(SO4)4 · 22H2O |
| 7.CB.85 | Redingtonite | (Fe2+,Mg,Ni)(Cr,Al)2(SO4)4 · 22H2O |
| 7.CB.85 | Wupatkiite | (Co,Mg,Ni)Al2(SO4)4 · 22H2O |
| 7.CB.90 | Meridianiite | MgSO4 · 11H2O |
Other Information
Notes:
Readily soluble in cold water.
Special Storage/
Display Requirements:
Display Requirements:
Dehydrates to chalcanthite. Virtually impossible to preserve in an artificial environment.
Health Risks:
No information on health risks for this material has been entered into the database. You should always treat mineral specimens with care.
Internet Links for Boothite
mindat.org URL:
https://www.mindat.org/min-720.html
Please feel free to link to this page.
Please feel free to link to this page.
Search Engines:
External Links:
References for Boothite
Reference List:
Localities for Boothite
Locality List
 - This locality has map coordinates listed.
- This locality has map coordinates listed.
 - This locality has estimated coordinates.
ⓘ - Click for references and further information on this occurrence.
? - Indicates mineral may be doubtful at this locality.
- This locality has estimated coordinates.
ⓘ - Click for references and further information on this occurrence.
? - Indicates mineral may be doubtful at this locality.
 - Good crystals or important locality for species.
- Good crystals or important locality for species.
 - World class for species or very significant.
(TL) - Type Locality for a valid mineral species.
(FRL) - First Recorded Locality for everything else (eg varieties).
- World class for species or very significant.
(TL) - Type Locality for a valid mineral species.
(FRL) - First Recorded Locality for everything else (eg varieties).
All localities listed without proper references should be considered as questionable.
Australia | |
| Leverett et al. (2004) +1 other reference |
| Ralph Bottrill et al in prep. +1 other reference |
| Bottrill et al. (2008) | |
Chile | |
| Hey +1 other reference |
France | |
| Coueille (1988) |
| Palache et al. (1951) +1 other reference |
Greece | |
| Rieck et al. (2022) |
Italy | |
| Scacchi (1872) +2 other references |
| Scacchi (1872) +1 other reference | |
| Exel (1987) |
Norway | |
| Garmo (1974) +2 other references |
| Ellingsen (1989) +1 other reference |
Russia | |
| maurice.strahlen.org (n.d.) |
USA | |
| Grant et al. (2005) |
| Mineralogical Record 11:225 +1 other reference |
| Brian Beck Collected |
| Van Nostrand Reinholt Press: 296. +5 other references |
| Mineralogical Record 20 129-142. | |
| Palache et al. (1951) |
| Van Nostrand Reinholt Press: 297 +6 other references | |
| |
| Murdoch (1966) |
| |
| Mineralogical Society of America - ... |
| Castor et al. (2004) |
| Rob Lavinsky |
| Finnell et al. (1963) |
| Finnell et al. (1963) +1 other reference |
Quick NavTopAbout BoothiteUnique IdentifiersIMA Classification Classification Mineral SymbolsPhysical Properties Optical Data Chemistry Crystallography X-Ray Powder DiffractionGeological EnvironmentType Occurrence SynonymsOther LanguagesVarietiesRelationshipsCommon AssociatesStrunz-MindatOther InformationInternet Links References Localities Locality List



 symbol to view information about a locality.
The
symbol to view information about a locality.
The 



Alma Mine, Leona Heights deposit, Leona Heights, Oakland, Oakland Hills, Alameda County, California, USA